As we’re uncovering more bits and pieces of data, we kept being lead to the idea that our membranes may be made out of SiO2 and have a modulus in the gigapascal range, but cells seem to perceive them as soft substrates. After looking at focal adhesions, fibronectin deposition, and cell/cell junction formation, we’re still sticking to the thought that the membranes are being interpreted by cells as soft substrates. To test this further we decided to look into the Hippo pathway and YAP (Yes-associated protein)/ TAZ (transcriptional co-activator with PDZ-binding motif) after reading several papers that looked at how YAP/TAZ plays a role in a cells mechanosensing.
For some background, YAP localizes in the nucleus when the cells are on a stiff substrate and it localizes in the cytoplasm when the cells are on soft substrates. To examine YAP on our membranes we wanted to set up the following experiment to again confirm that the cells are behaving similarly on our membranes as soft substrates.
For the substrates we wanted to use 0.5μm HP, 3μm HP (old), 3μm HP (new), TCP, and nonporous SiO2. The last time we buil the devices, the “new” 3μm HP membranes that we just got seemed to be more fragile than the membranes we have been using so we decided to run both of them just to see if the new ones were more prone to breaking. It turns out that none of the 3μm membranes broke over the course of the experiment.
For the experimental methods, cyto-Vu devices were made using the above substrates and 600 HUVECs were seeded per membrane. For the ADSC trials, 400 ADSCs were seeded per membrane. The cells were allowed to adhere to the substrate for 1 hour and then the wells were flooded with 800uL of media. After 3 days, the cells were fixed in 3.7% formaldehyde for 15 minutes, permeabilized with 0.1% Triton T-100 for 3 minutes, washed with 4% BSA for 15 minutes, and 100uL of the YAP antibody (0.2mg/mL was diluted 1:100 in PBS) was added ontop of the substrate for half an hour.
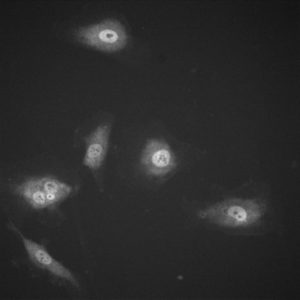
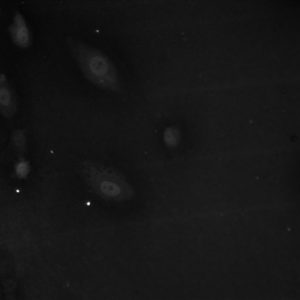
The images were acquired and analyzed in image j. The nucleus’ brightness was measured as well as the background and the cytoplasm of the cell. To analyze the data, the background was subtracted from the values for the nucleus and the cytoplasm. Then, the intensity of the nucleus was divided by the intensity of the cytoplasm. Sample sizes were at least 20 cells. The results are as follows:
For HUVECs:
0.5μm HP: 1.99 Standard deviation: 0.41
3μm HP: 1.40 Standard deviation: 0.35
TCP: 2.18 Standard deviation: 0.61
Nonporous SiO2: 2.22 Standard deviation: 0.28
The images of the ADSCs showed little to no nuclear staining in all cases so I’m going to re-stain the plates that I have for one hour to see if that was the issue.
The images below show how the YAP/TAZ localizes on the stiffer substrate but the ratio of nucleus intensity to cytoplasm intensity decreases on the porous substrates.
Discussion:
Here is another aspect that we have looked at t see if our membranes are interpreted by cells as soft substrates and in this case, it falls into line with our previous experiments. Although more cells are available for analysis and statistics haven’t been run yet, results are promising. However, with the 3μm HP membranes the stain didn’t seem to take as well and although data could be garnered it would be of merit to confirm the results another time. The localization shows that the membranes exhibit not as much nuclear YAP as the stiff control substrates indicating the cell sees them as a soft substrate. It would be interesting to compare these numbers to the silicones we have tested as a baseline. Also, blocking YAP might be interesting to see if we still observe the same phenomena in regards to focal adhesions, fibronectin, and junction proteins. Results for ADSCs will follow in the morning!
Spencer