Finalizing Microfluidic Shear Calculations
Introduction
As a continuation of my rotation project and a beginning to my PhD work in the McGrath lab, I wanted to finalize my shear stress calculations within our blood vessel microfluidic device. All previous shear stress calculations were purely analytical, with many assumptions. This work aimed to minimize those assumptions while providing some publishable data to ensure our readers (or users) that the shear stress within our blood vessel model is in fact accurate. The compliant nature of the PDMS, silicone gaskets, and collagen gel may lead to bending in the case of fluid flow. This bending leads to increases in channel size that, within the micron scale, can lead to major variability in flow and thus shear stress. One way to ensure this compliance is not effecting our shear stress is to measure it in situ. One established way to perform this measurement is to track micron sized fluorescent beads within the device channel and map their trajectories (Galie et al., Lab on a Chip 2015). The analytical procedure behind this process will be described in detail below.
Navier-Stokes and Shear Stress
Beginning with the equations for flow of a in-compressible Newtonian fluid through a rectangular channel (Navier-Stokes Cartesian Coordinates), we can derive an equation for the fluid velocity profile within our blood vessel device.
Assuming steady, fully developed, laminar flow, negligible gravity effects, smooth surfaces and symmetry, the equation for flow through the rectangular channel is:
The PDE can be solved for flow through the channel, however, this derivation involves a non-finite component and further assumptions that only add a slight increase in accuracy:
(First equation from Microbiomechanics class materials, Jiandi Wan)
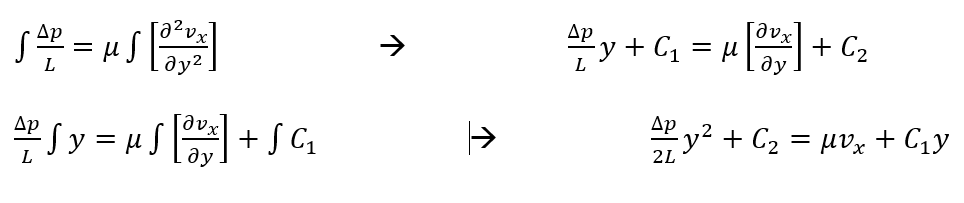
The PDE can be greatly simplified, however, if we assume flow between parallel plates instead:
Solving this equation for the velocity profile of the laminar fluid flow we get:
Using the definition for volumetric flow rate and our derived velocity profile, we can solve for flow between the plates (Hagen-Poiseuille equation):
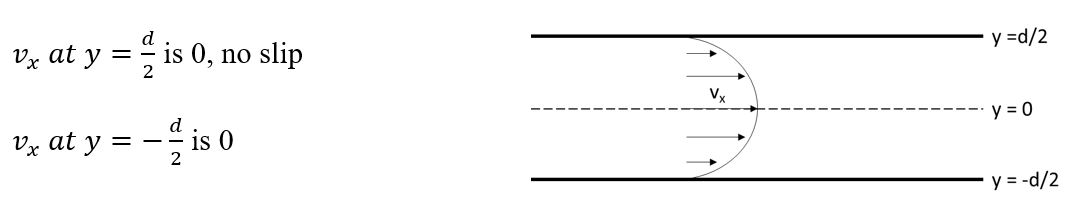
Now we can solve for wall shear stress within our device (using Newton’s Law of Viscosity) in terms of Q and maximum velocity.
At the wall, y =-d/2
Velocity is maximum at y=0
Fluorescent Bead Velocimetry
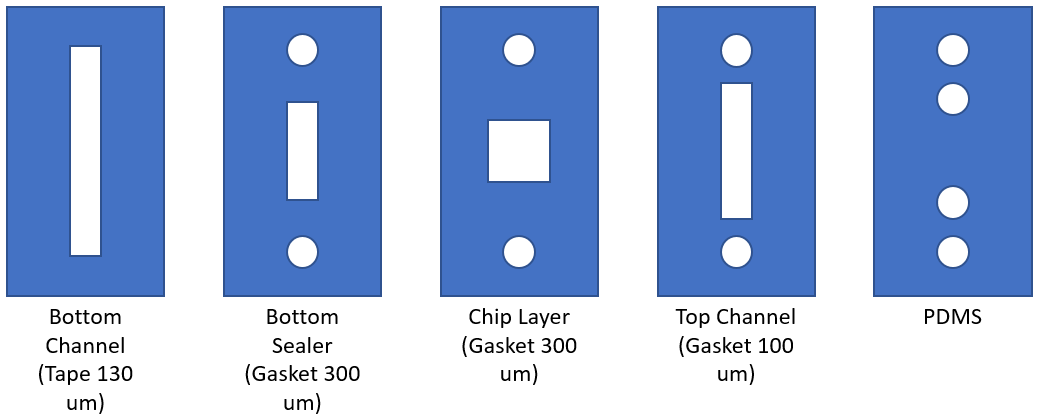
The blood vessel device includes a 130 um (d) by 2 mm (w) rectangular channel with a rat tail collagen gel on the bottom and PDMS on the top. The entirety of the device design can be seen in Figure 1. 1 micron fluorescent polystyrene beads were incubated in HUVEC media (MCDB-131) overnight to prevent bead adherence to the collagen gel. A peristaltic pump was connected to a capacitor and reservoir with the top channel of the device connected in-between (Figure 2). The pump flow rate was set to 6.5 x 10^-4 cm^3/s and the bead/media solution was added to the reservoir. The assembly was placed under the fluorescent microscope and the microscope was focused at the midpoint of the device (i.e. 65 um above the chip surface). Videos were recorded at 30 fps at 20x.

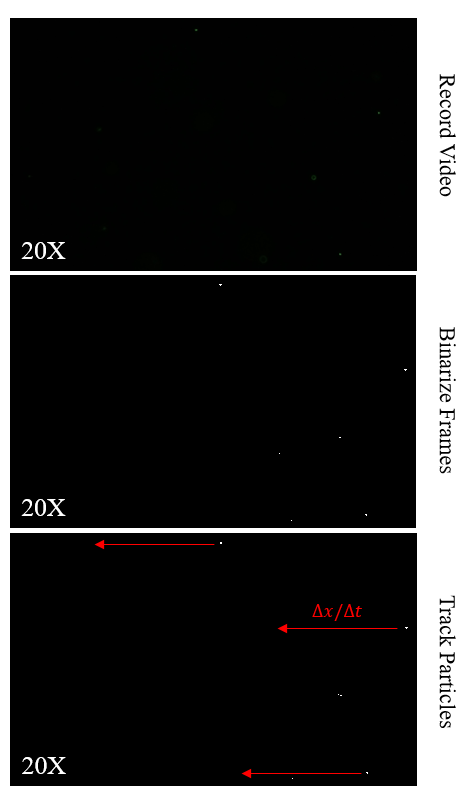
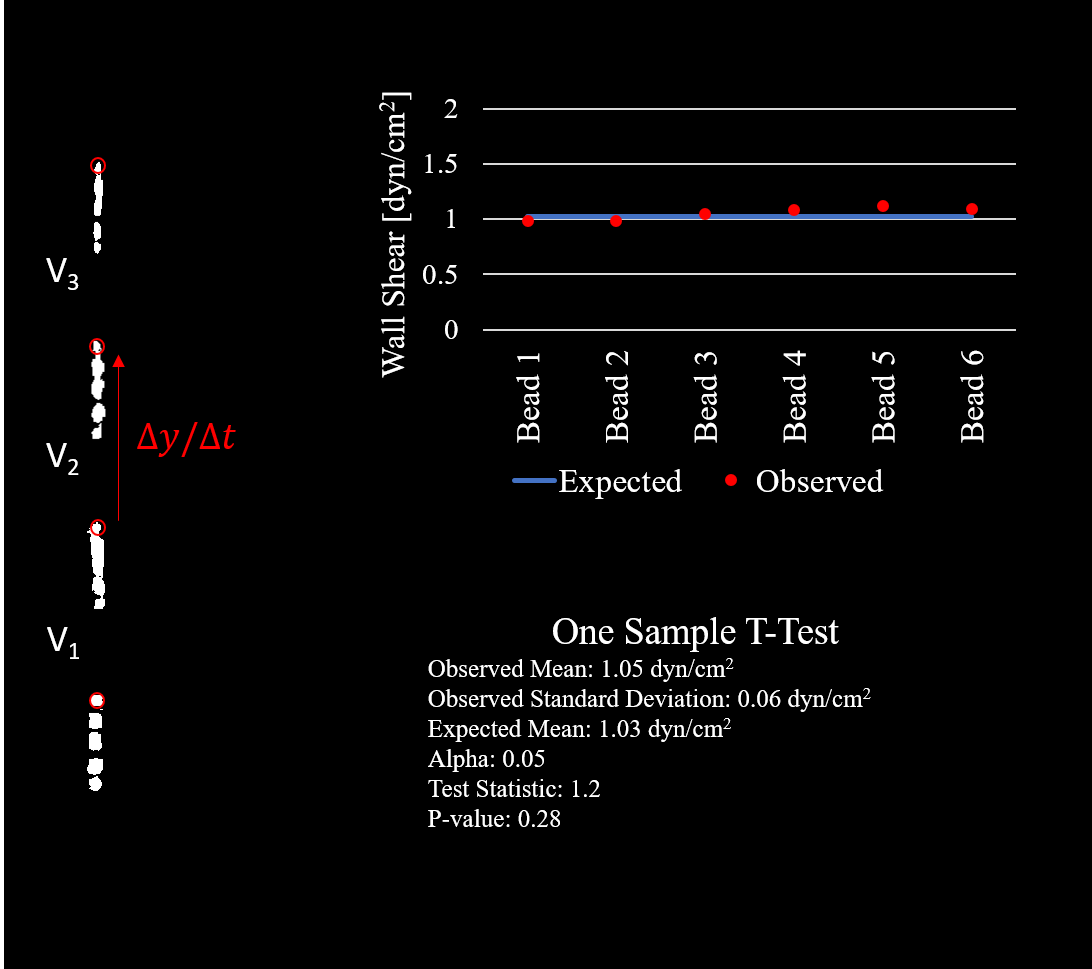
Post experiment tracking was performed in matlab using a simple code for importing, binarizing, labeling, and tracking of the beads over the course of about 5 to 6 frames (Figure 3). The expected wall shear stress from the pump flow rate and derived equation for shear at the wall in terms of Q is equal to 1.0269 dyn/cm^2. Tracking of 6 beads gave me a value of 1.05 +/- 0.056 dyn/cm^2 (average +/- standard deviation).
Conclusion
Statistical analysis (One sample t-test) fails to show any significant difference between the observed and calculated means (Figure 4). Therefore we can have confidence in saying the walls of our microfluidic device (and thus the HUVECs) are experiencing a degree of shear very close to what we calculate based on pump speed. The imaging capabilities limited our ability to track particles at the speed they travel at 12 dyn/cm^2 (the shear we aim to achieve in our device). For this reason, we are extrapolating our results from a lower pump speed. It is possible this small degree of error (though not significantly different) may increase with increased flow rate. If this issue is brought up in paper review (or user review) we can drop the necessary flow rate to achieve the same shear by increasing medium viscosity (dextran is an option), otherwise we can aim to track particles at the full velocity with a nicer camera with a very high frame rate.


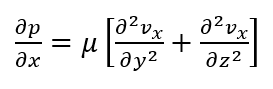

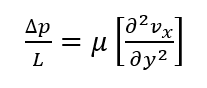
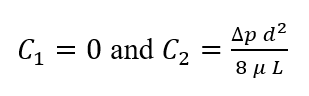
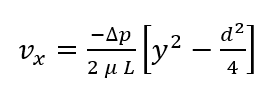
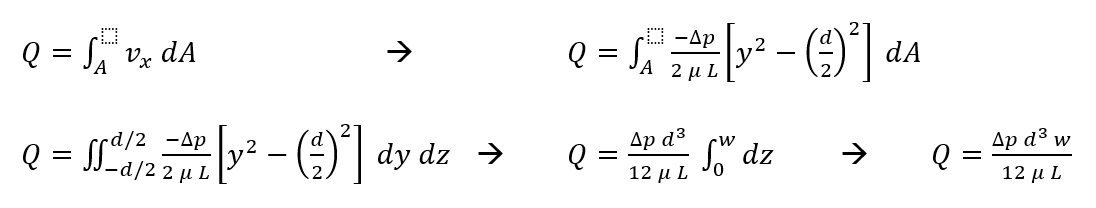
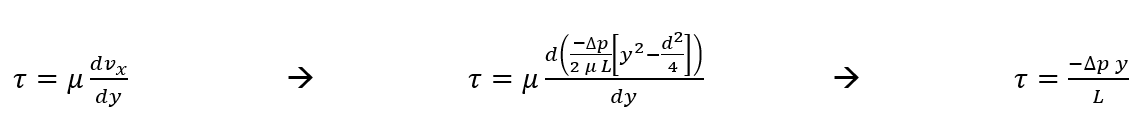
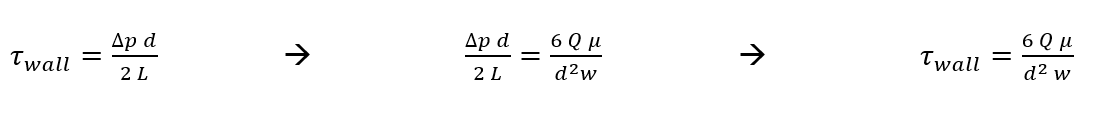
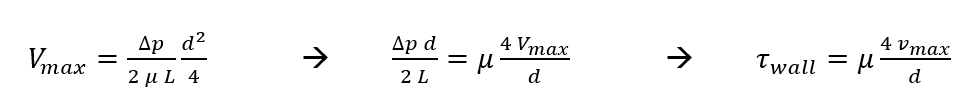
nice posts. cool device pic!