Brain Extract Separations using Small Scale EQ
The new smaller EQ setup that Tom came up with were tested in these experiments. The benefit is that there is much less dilution of our filtrates. 500ul of our sample (in these cases we tested 1.0 mg/mL and 0.5 mg/mL brain extract) is loaded into the SepCon and 250uL of buffer fills the reservoir under the SepCon (opposed to the 500uL reservoir of previous EQ setups).
I think this set of experiments illustrates the current weakness of the membranes. Out of 12 samples from w340, 1 survived and passed ~194uL after 5.75 hours. Out of 4 from w341, 1 survived and passed ~30uL after 4 hours. None survived from 6 w342 trials. The starting sample in both cases was .5 mg/mL brain extract. The filtrates were concentrated close to actual volume passed using 3k Nanoseps and run on a 7% gel.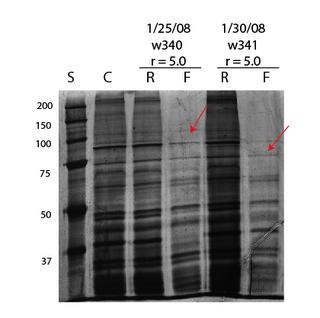 It looks that we are getting brain extract separations using the SepCons as before. We are apparently still not passing enough material to see reduction of lower molecular weight species in the retentate.
It looks that we are getting brain extract separations using the SepCons as before. We are apparently still not passing enough material to see reduction of lower molecular weight species in the retentate.
In regard to my previous post about depleted serum separations, maybe it’s the mixture in question that’s not giving us a separation. I think a good step would be to perform the same diffusion experiments with the membranes used for the serum separations, but use brain extract this time. We’ve always been pretty lucky to see separations using brain extract, but this may just be a function of the mixture and what complexes it contains.
Is the retentate diluted back to the original volume before the gel is run? If not, we should never see any depletion of the lower molecular weight species, correct? Of course, if you dilute the retentate, you won’t see concentration of the higher MW species. I guess it all depends on what you want to see…