eLIFE submission: A silicon nanomembrane-based in vitro platform to visualize immune cell trafficking across the human blood-brain barrier
We have been collaborating with Britta Engelhardt’s group at the University of Bern in Switzerland since Tejas and I met them at the CNS Barriers conference in Paris in 2015. They are interested in studying live T-cell transmigration across human BBB models as part of their research program in Multiple Sclerosis (MS). Prior to working with us, they spent many years trying to develop technologies for both the cellular and device aspects of a human BBB model. On the cellular front, they were part of a team that introduced “brain-like endothelial cells” or BLECs (1). BLECs are derived from umbilical vein CD34+ hematopoetic stem cells that, when co-cultured with pericytes, produce tight barriers and other BBB characteristics.
Britta’s group argues that flow is essential for proper T-cell diapedesis (2-4). In trying to make dual chamber (i.e. transwell) flow cells for their work with BLECs, they tried many membranes and found that that the imaging was bad and the adhesion was often poor. The manuscript includes a long list of materials that didn’t work (see the supplement). Fortunately for us, NPN works great.
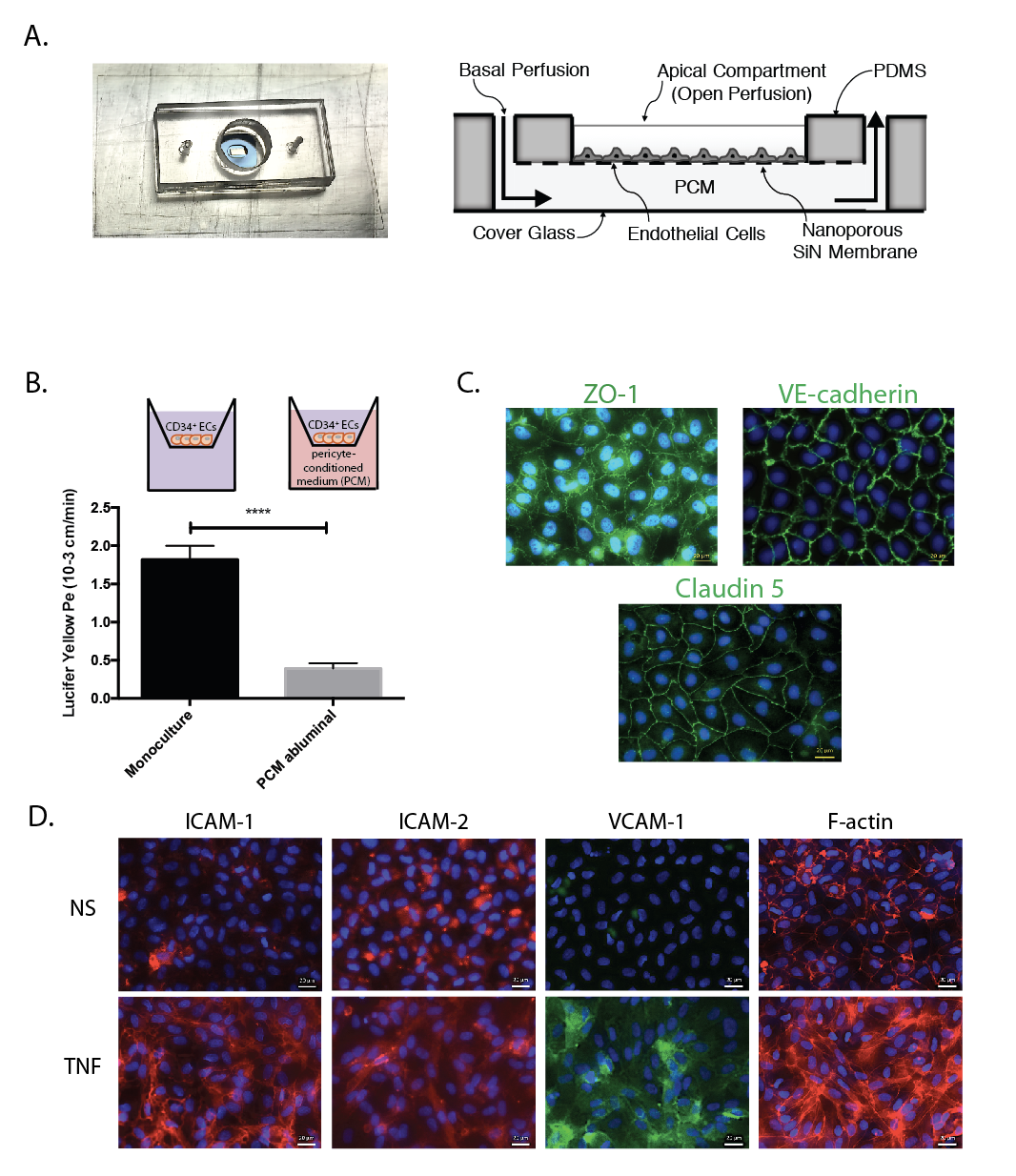
The manuscript suggests that our NPN-based culture devices and BLECs are the ideal human BBB platform, particularly for the study of T-cell transmigration. It certainly validates NPN as a substrate for BBB models by showing all the key junction markers and low permeability to small molecule diffusion. One of the key innovations in the paper is the ability to replace the pericyte co-culture with pericyte condition media (PCM) added to the bottom chamber. This is an enormous simplification and avoids the problem of having two cell layers in focus when imaging. The fact that addition of PCM lowers barrier properties when added to the bottom chamber, but not when added to the top chamber alone, is evidence of the polarization of BLECs in transwell-style systems. In fact this is the most direct evidence I’ve seen that the culture on a permeable membrane is necessary to induce barrier phenotypes.
I’ve included the validation figure (done in our ‘transwell mimetic’ devices) and the movies here. The movies are the crown jewels of this project. The complete manuscript is here: Musso(Engelhardt)submitted
References
1. Cecchelli, R.; Aday, S.; Sevin, E.; Almeida, C.; Culot, M.; Dehouck, L.; Coisne, C.; Engelhardt, B.; Dehouck, M. P.; Ferreira, L., A stable and reproducible human blood-brain barrier model derived from hematopoietic stem cells. PloS one 2014, 9 (6), e99733. -> Cecchelli(Ferreira)PLoSOne14
2. Cinamon, G.; Shinder, V.; Alon, R., Shear forces promote lymphocyte migration across vascular endothelium bearing apical chemokines. Nature immunology 2001, 2 (6), 515-22.
3. Schreiber, T. H.; Shinder, V.; Cain, D. W.; Alon, R.; Sackstein, R., Shear flow-dependent integration of apical and subendothelial chemokines in T-cell transmigration: implications for locomotion and the multistep paradigm. Blood 2007, 109 (4), 1381-6.
4. Steiner, O.; Coisne, C.; Engelhardt, B.; Lyck, R., Comparison of immortalized bEnd5 and primary mouse brain microvascular endothelial cells as in vitro blood-brain barrier models for the study of T cell extravasation. Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism 2011, 31 (1), 315-27.