Microfluidic debubbler
What is it?
A device to remove bubbles from microfluidic lines. Air and liquid enter on one side and only the liquid leaves. Our debubbler is disposable, cheap (<$0.50) and easy to make in the lab.
Why use a debubbler?
Bubbles can have a severe negative effect on cells cultured in a microfluidic chamber. The bubble blocks the channel, decreasing the height of the fluid channel and greatly increasing the shear force on the cells. This impacts cell viability and can even strip the cells off the culture substrate. An in-line debubbler removes these bubbles and solves these problems.
How is the device made?
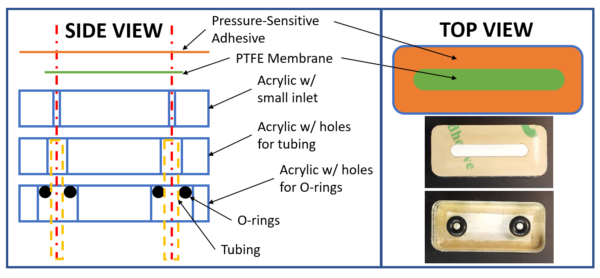
Our device is made of three layers of laser-cut acrylic, a PTFE microporous membrane, and a piece of pressure-sensitive adhesive. The bottom layer of acrylic has holes to accept O-rings to seal the tubing in place. The second layer of acrylic has holes that fit around the tubes, so they can be pushed in a couple of millimeters past the O-rings. The third layer of acrylic has small holes to allow fluid to pass through to the membrane but prevents the tubing from puncturing the membrane. The PTFE membrane sits directly on top of this third piece of acrylic and is sealed in place with the pressure sensitive adhesive.
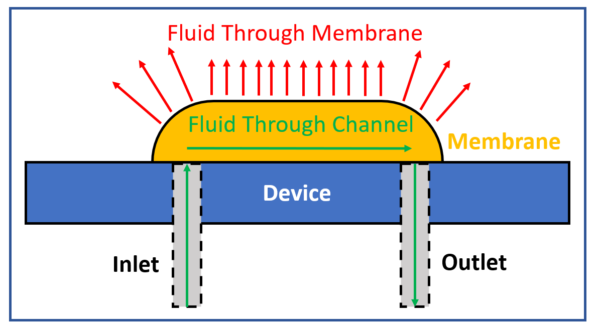
How does it work?
Syringe pumps flow both air and liquid at constant rates, which combine at a wye junction and then flow into the bubble trap. This generates a pressure inside the bubble trap between the acrylic and Teflon membrane. This pressure forces the air through the pores in the membrane, however the water cannot pass because the membrane is hydrophobic. The air moves across the membrane and through the membrane at different rates. If the air diffuses across the membrane before it reaches the outlet of the bubble trap, the device will function as intended and completely remove air from the line.
I have noticed that the device will function until the membrane breaks, as evidenced by liquid appearing on the other side of the membrane. Therefore, there is a maximum pressure the device can withstand before breaking.
MATLAB Analysis:
First, the .mp4 video file is loaded into MATLAB and split into a series of individual frames. Each frame is cropped to specific markers on the background that were measured to be 75mm apart. This allows me to convert from pixels to millimeters. The frames are then converted from RGB color to grayscale, so the image can be binarized with a threshold. After some filtering, the liquid portions of the fluid stream appear as separate objects. The code then finds the ends of each of these objects, so some metrics can be calculated. With these markers, I can calculate the size of each bubble and the ratio of air to liquid in the fluid stream. The code then compares how far the first marker has traveled from frame to frame and uses this information along with the frame rate and tube dimensions to calculate an average flow rate. My code takes a video input and calculates an average bubble size, average bubble density, and average flow rate.
Results:
My device removes bubbles ranging from 0.7μL to 2.5μL at a bubble density ranging from 40% to 70% at flow rates between 0.17mL/min to approximately 8mL/min.
According to Alec’s equation for shear stress at the wall of a microfluidic channel, these flow rates correspond to 15 dyne/cm^2 and 700 dyne/cm^2.
Matt – please add debubbling videos and update post once cell culture media and long term operation experiments are compete