Small Particles Task Force: Observation of Web-Like Structures on Microslit Membranes Following Tween-20–Assisted Microparticle Dispersion
Introduction
The LOMP Small Particle Taskforce is focused on developing a reliable and rigorous procedure for analyzing small microplastics in environmental samples. Our primary aim is to ensure that the data we generate accurately reflects the true microplastic population present in each sample. Achieving this requires careful attention to representativeness, verification, and contamination control throughout the entire workflow.
Three core objectives guide this effort. First, we must confirm that each sample analyzed is truly representative of the broader environmental system we are studying. Second, it is essential to verify that the particles counted are indeed microplastics, supported by appropriate analytical methods. Third, minimizing contamination from collection to processing to analysis is crucial for ensuring confidence in the final results.
To date, we have made significant progress in reducing methodological contamination and have begun refining our staining approach. However, further work is needed to better remove environmental contaminants and to verify that the particles we measure accurately represent the true population. This next phase of development will involve benchmarking to strengthen both reliability and reproducibility.
To better align the analytical method across LOMP, the MMC arm of the Small Particles Taskforce made stocks that all groups were asked to analyze with their current methods. This post walks through the process of preparing stock and analytical methods by the groups.
In a previous blog post titled SPTF – Initial Stock Preparation & Staining Workflow, we discussed the preparation of the stock that the MMC created for the SPTF for analysis.
Results & Discussion
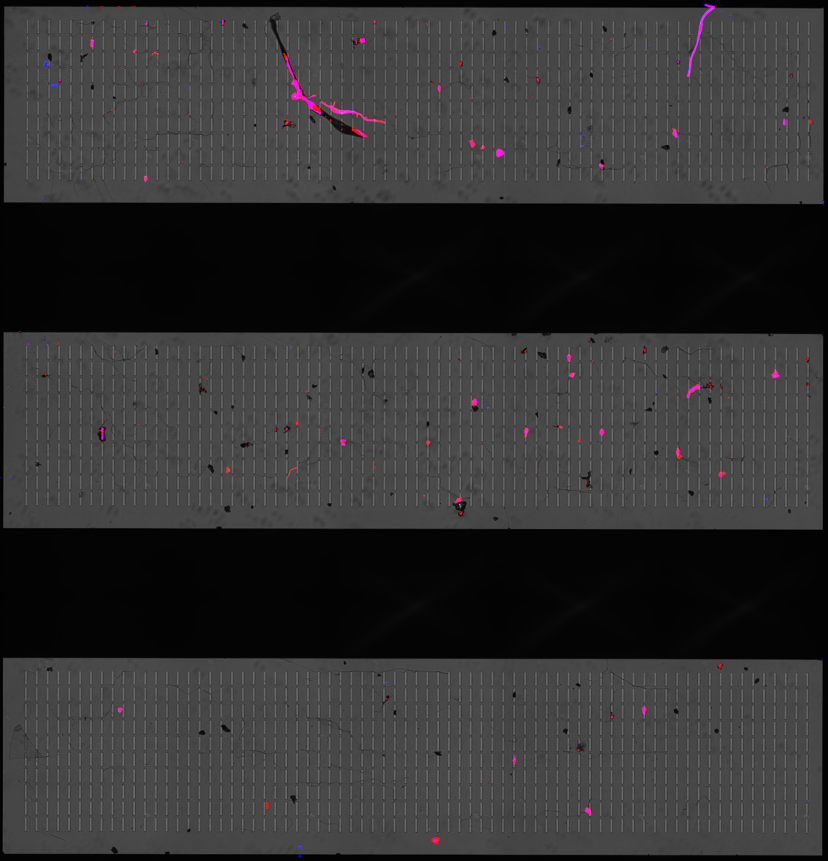
Figure 1 below illustrates the capture of MMC cryomilled particles on an 8 µm membrane. Figure 1a was 0.5 mL of the solution filtered directly and stained. Particle coagulating together, as shown in Figure 1a below, especially at high volumes and concentrations of particles, is a common observation when filtering to count 1) the size of particles and 2) to enumerate individual particles for concentration and count calculations. To better isolate individually captured particles, 0.1% Tween-20 was used as a surfactant to avoid particle clumping for the samples in Figures 1b-c. The addition of the Tween-2o seemed to help with particle distribution across the 3 membrane windows, and particles visually look better separated.
 |
 |
 |
 |
| a | b | c | d |
| Figure 1: MMC Spiked particles filtered through a microslit membrane. Where a) is 0.5 mL filtered without Tween-20, b) is 0.5 mL filtered with 0.1% Tween-20, and c & d) is 1 mL filtered with 0.1% Tween-20. | |||
Despite Tween-20 giving better individual particle separation, all three Tween-containing samples showed a faint web-like pattern left behind on the membrane surface. This pattern wasn’t present in the samples without Tween. The origin isn’t fully clear, but as shown in Figure 2b, the structure does pick up Nile Red, which suggests it’s an inorganic material rather than membrane damage. The most likely explanation is that this is a drying artifact from residual Tween-20. When Tween dries on a flat, smooth surface like the microslit membrane, it can form a very thin film that contracts as water evaporates. As that film pulls inward, it can leave behind these crack- or web-like lines that show up in both brightfield and fluorescence. In other words, the “web” is probably not particles or biological material, but just leftover surfactant drying in a patterned way on the membrane. An example of this drying pattern of various surfactants is at in a paper by Shi, et al (Langmuir 2021, 37, 14, 4091–4101). Figures 3 and 4 show two figures from the Shi et al. paper illustrating this drying phenomenon and comparing it to the drying of pure water. I would recommend using isopropyl alcohol, which I have written about in a different blog post found here.
 |
 |
 |
| a | b | c |
| Figure 2: Images from MMC Spiked samples with Tween-20 showing web-like structure on membranes. | ||
| Figure 3: Bottom-view images during the drying of CnEm droplets. The images here only show the fully dried samples |
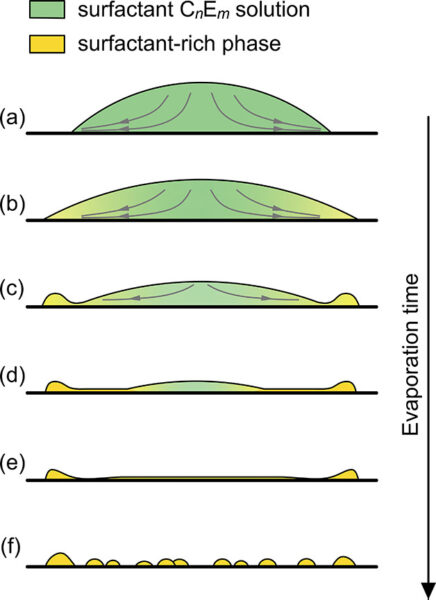 Figure 4: . Illustration of the drying process of CnEm dissolved droplets. The scale in the sketches do not reflect real scales.
Figure 4: . Illustration of the drying process of CnEm dissolved droplets. The scale in the sketches do not reflect real scales.