Wetting of Nanocavities using Isobaric Cooling
Summary and Instructional video
The simplest way to get something wet is to allow water to condense on the surface of the something. In our case, we have a number of nanocavities (~ 0.1 femtoliters) with porous tents over them. The easiest thing to do is simply chill the substrates down to allow condensation to form. If the condensation forms on all surfaces, it will wet the inner surface of the nanocavity as well as the outer portion. We have video evidence of Karl blowing on the backside of a nanomembrane to allow water to flow through it, due to a little backside pressure (5.5 PSI); once the backside surface is wetted, the actual hydraulic resistance is small.
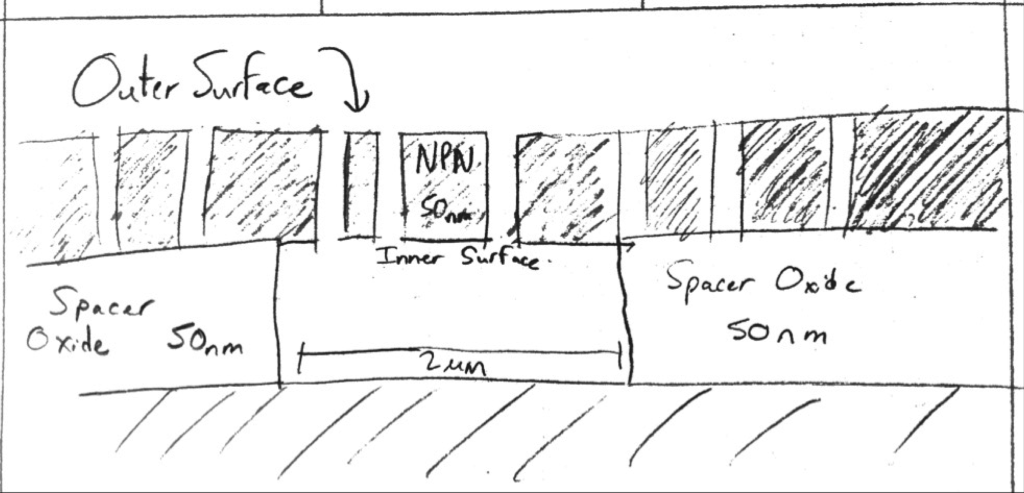
Therefore, it seems reasonable that the condensation will break the backside surface tension and allow condensate that forms on the outer surface to flow inward (0.1 femtoliters).
I tented a few structures (Wafer 1153, went through ozone compatible RTP process, 700 C, 5 min), as well as used some previously tented materials (SEM samples found here). To cool the surface, I placed all the samples in a gel box, closed the box, and put it in the -10 C freezer for 15 min. I then removed the box, opened it, and examined it under a microscope at 20x and 40x, capturing video after ~0.5-1 minute of finding the active area.
State A – ~Room Temperature
- 1 atm
- 52 degrees F = 11 degrees C = 284 K
- Ideally 297K
- 3.16 kPa vapor pressure of water
- RH 20-30% (39% @ 52C in Rochester NY, 4/21/2015)
State B – Freezer
- 1 atm
- -10 degrees C = 263.15 K
- < 0.611 kPa vapor pressure of water @ 0 degrees C
- Condensate Forms
State C – Room
- 1 atm
- 52 degrees F = 11 degrees C = 284 K
- 3.16 kPa vapor pressure of water
- RH 20-30%
In each of the videos, you can see condensate microdroplets disappearing from the outer surface and changing the color of the local holes underneath to a reddish color, which is weak evidence indicating that the water is flowing down into the cavity (refractive index change of water thin film). As the membrane dries (in the second video), each of the holes dries out independently, which is weak evidence for separate holes fluidically.
The strategy for wetting a device is therefore:
- Incubate box in humid room temperature air
- Place samples in box
- Cool down in freezer
- Remove and add additional water after 1 minute (to allow pores to finish wetting)
- Use device or store wet.
If above is borne out, we don’t even have to wet both surfaces of SEPCONs any more; just chill it. No need for wetting both surfaces of a nanomembrane tangentially; just chill it if you want a flow through the membrane. The key will be controlling the humidification and cooling processes.
Outstanding questions:
1. What does a control surface look like (no freezing step, instead heated in 70C oven for 10 minutes, evaporating residual water)?

- I dropped 5 microliters to wet the outer membrane surface, then removed as much water as I could from the bubble to get a quick evaporation answer (3 minutes start to finish)
This is an interesting video that bears further discussion. It looks like all the pores are wetted, but they don’t evaporate as cleanly as the chilled ones. Is it due to the water being introduced to the hot chip (make condensate inside each femtoliter volume)?
2. Do the nanomembranes droop down and contact the surface of each hole during wetting or drying?
- Probably during drying. Unsure about wetting. We can use the SEM to find out.
super cool! You have Newton’s colours in these images, one could calculate the film thickness thereby, and by plotting the time series even calculate the kinetics of returning to equilibrium. Can you increase the deposition by doing this under pressure?
https://www.filmetrics.com/reflectance-calculator
I’m going to need some more precise measurements of thickness of the different films (SEM), but then we can calculate based on the different regions (open holes, wetted holes, water on top etc). The film stack is actually complicated (4 layers, similar thicknesses, different indicies), so thickness differences could well throw off the proper color.
What do you mean about increasing the deposition? Increasing the amount of hydration on the surface? Probably. Additionally, there’s surely an optimal wetting bubble size; I think there may be a tradeoff between condensation inside the cavity, and forming larger bubbles on the surface. What if the outer surface condensation dehydrates the inner surface through surface tension? Like you said, very interesting kinetics to find equilibrium.