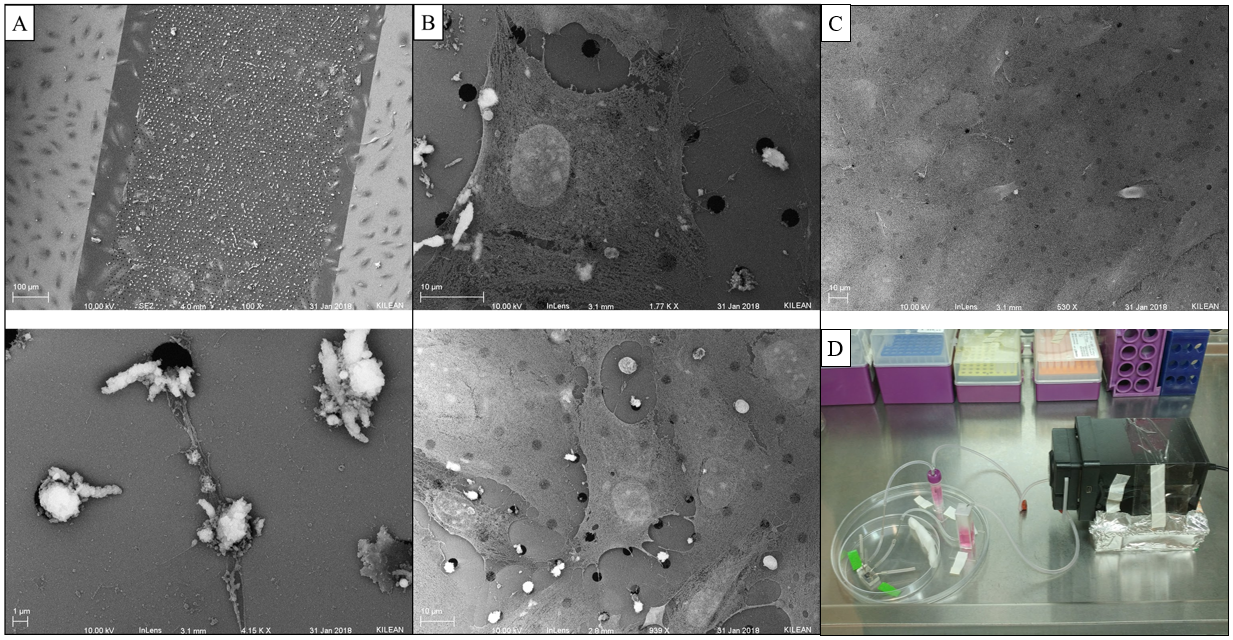
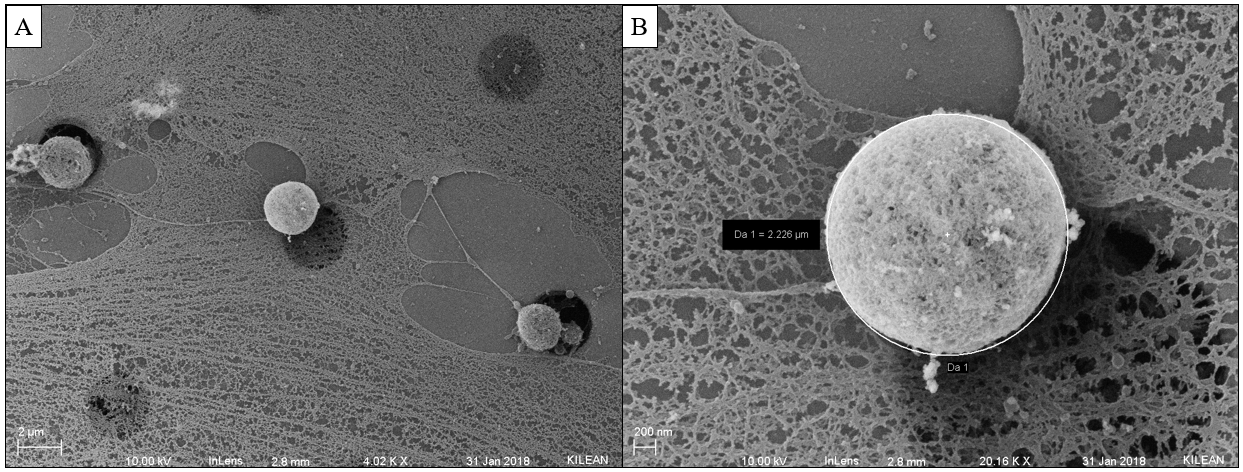
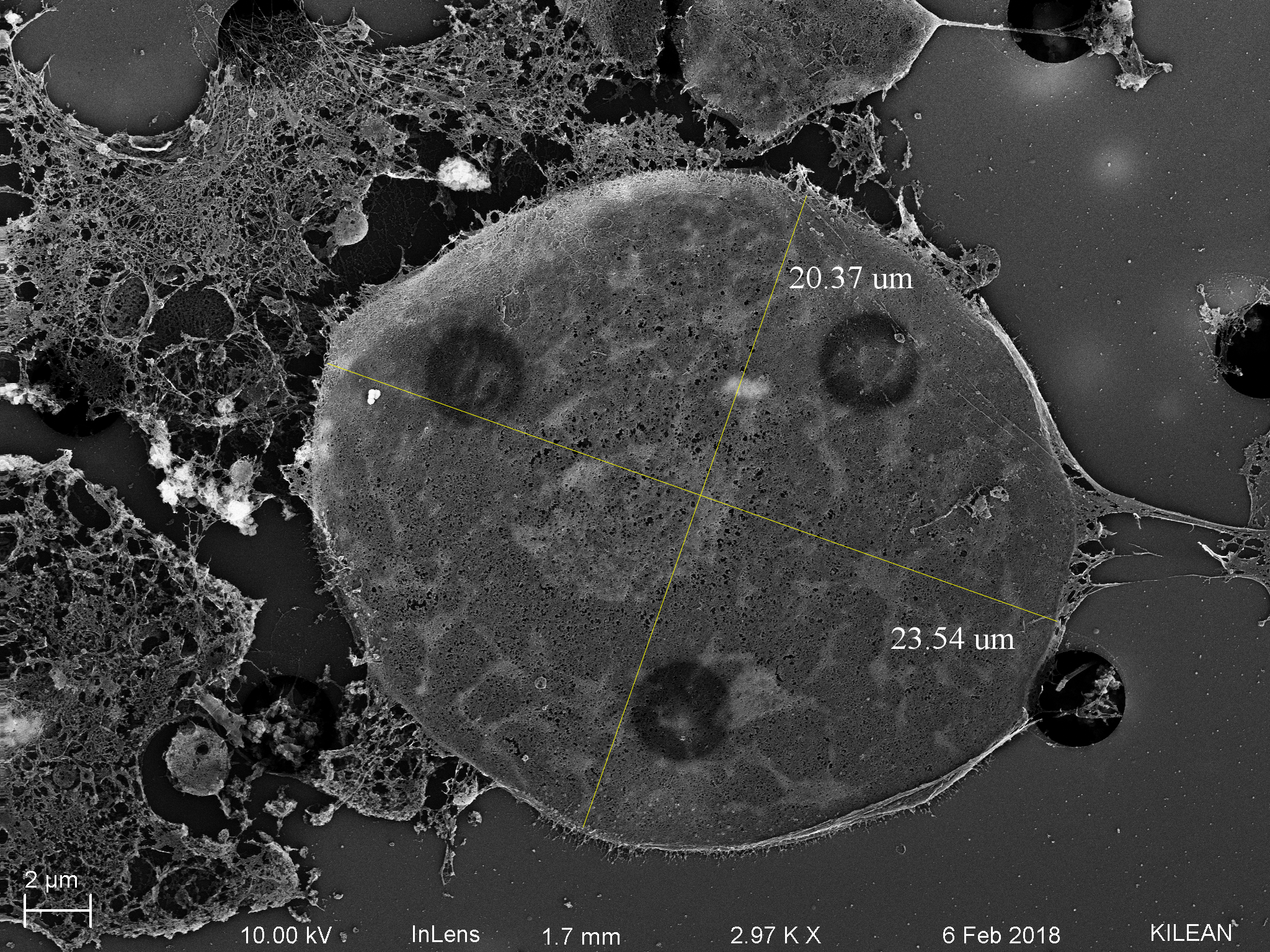
SEM of Sheared/Non-sheared HUVECs
Motivation and Procedure
After discovering that pores on 3um LP SiO2 membranes seem to get blocked following 3 hours of shear [of HUVECs], Kilean and I decided to utilized SEM to further inspect what might be doing the occluding. HUVECs were seeded onto the membranes as usual and left to grow to confluent for 24 hours. Devices were then attached to a flow system and sheared at 10 dyn/cm^2 for 3 hours, 1 hour, or not at all (static control). Cells were fixed immediately in 2.5% glutaraldehyde for 5 mins at 37 C. The PDMS was removed from the device and serial dehydration was performed in 10 min increments of 50%, 60%, 70%, 80%, 90%, and 100% EtOH. The membrane was extracted from the device in 100% EtOH (Ethanol facilitated disassociation of the 3M tape from the membrane). Membranes were dried via CPD to preserve cell structure and coated with 7 nm of platinum.

TMP Balanced Flow
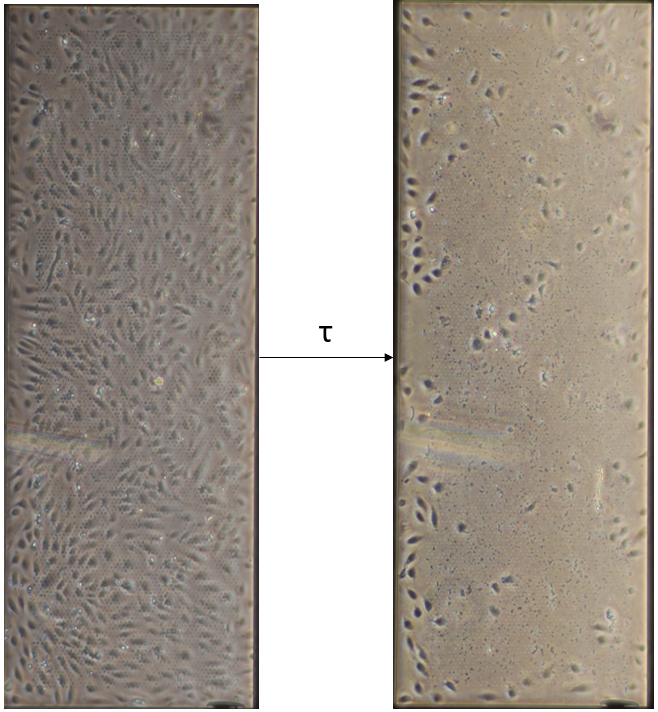
One major suspicion of the pore occlusion phenomena is that transmembrane pressure drives cell debris into the pores. The bottom channel of a standard culture device was closed (Figure 5) to prevent any fluid exchange between channels. After filling the bottom channel and connecting the tubing, cells were seeded and left to grow for 24 hours as usual.

Conclusion(s)
Cell debris is most likely the cause of pore plugging we see with or without flow (can confirm with ICC for actin within the pores). This phenomenon is not entirely pressure driven, so it is most likely a cell function driving protrusions into the pores, potentially influenced by flow. It is also important to note that cell nuclei have the ability to translocate through 3 um pores, pointing to their extreme flexibility.