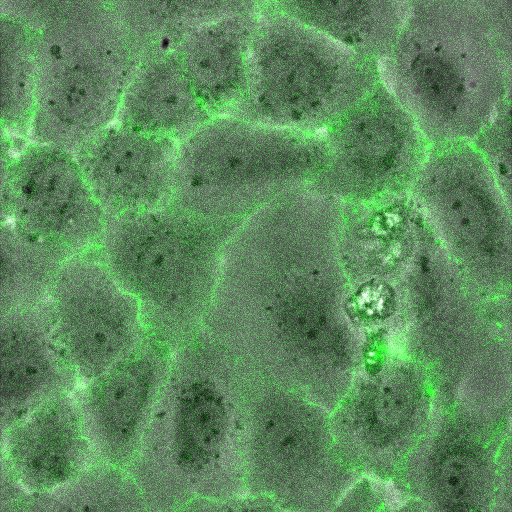
Effects of Luminal and Abluminal TNF-α on Neutrophil Migration
Introduction
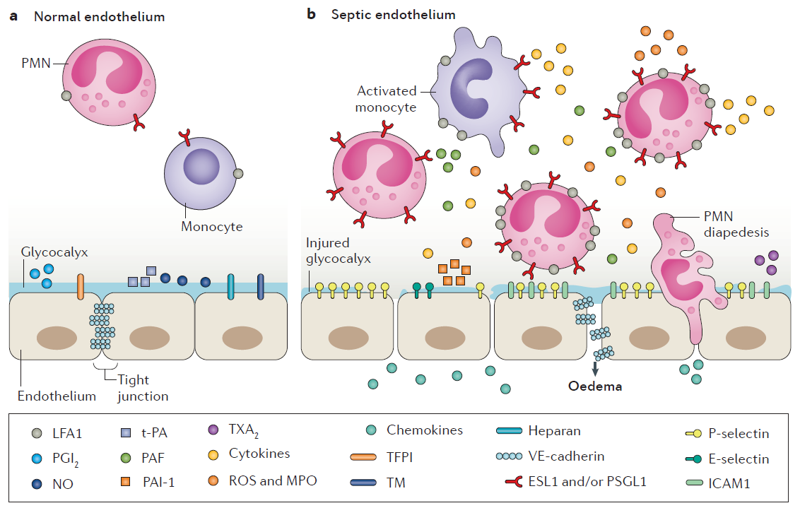
In severe sepsis, a primary infection gives way to a maladaptive immune response leading to wide spread edema and multiple organ dysfunction. The initial infection can occur in many peripheral tissues throughout the body. The pathogen associated molecular patterns (e.g. LPS on gram-negative bacteria) interact with pattern recognizing receptors on resident macrophages and endothelial cells (e.g. LPS stimulates endothelial cell toll-like receptor 4). This signaling cascade results in increased cytokine and adhesion molecule production, which in turn further stimulates circulating immune cells and endothelial cells, leading to targeted neutrophil migration, reactive oxygen species production, and endothelial cell barrier disruption (Figure 1). If the infection goes untreated, or persists through the initial immune response, the cytokine production can spread throughout the vasculature, leading to nonspecific immune cell egress and barrier damage. With our optically favorable vascular mimetic, we would like to study the differences seen between the primary infection response and the wide spread response. Preliminaily, we would like to explore the migratory patterns of neutrophils on/through endothelial cell monolayers stimulated either luminally or abluminally with TNF-α.
Methods
Human umbilical vein endothelial cells (HUVECs) were seeded on 75 nm NPN membranes (1275) and left to adhere for 4 h. Media was exchanged luminally or abluminally with 20 ng/ml TNF-α in MCDB-131 and left overnight (16 h). Control devices had media changed with clean MCDB-131. Neutrophils were isolated from healthy donors the morning of experiments and used within 4 h of isolation. Devices were removed from the incubator and rinsed with fresh MCDB-131. Neutrophils were added to MCDB-131 at a density of 4.5 million cells/ml and perfused into the luminal channel of the vascular mimetic. Time lapse videos were recorded on a heated stage to maintain physiological temperature.
Results
Negative Control: Clean MCDB-131
Positive Control: Clean MCDB-131 followed by 10 nM fMLP stimulation abluminally
Luminal TNF-α Stimulation
Abluminal TNF-α Stimulation
Total TNF-α Stimulation
Discussion
With n=2, it appears that luminal TNF-α may be insufficient to induce a diapedisis event, where as abluminal TNF-α is. Importantly, the device is washed pre-neutrophil addition in an attempt to eliminate effects of TNF-α interacting with the neutrophils themselves. Of course, it is possible the wash is insufficient and the differences seen here may be due to residual TNF-α. Interestingly, the total TNF-α stimualtion devices did not appear to permit transmigration. I am still working on formulating a hypothesis around these subjective conclusions, but one tool that may help us moving forward is a collaboration with post-doc Jeff Tithof in Doug Kelley’s particle tracking lab. He has shown some potential to track luminal crawling, transmigration events, and abluminal crawling, giving us the ability to monitor a wide range of migration habits.
In the meantime, our next best bet is to explore possible mechanisms involved in this polar signaling response. I have been working on live cell ve-cadherin staining that may help with effort. I also hope to explore other contenders in the transmigration cascade including ICAM and p-selectin.