KMnO4 as a Platform: absorbance vs. concentration
I discussed with Jess the possibility of using a negatively charged dye. People did use such dyes before but we probably need to order them at this time. Since now we can use the spectrophotometer to determine the concentration of a solution, we might be able to make use of the potassium permanganate (KMnO4) solution we prepared before.
I have done a group of serial dilutions of KMnO4 to find the absorbance-concentration characterization curve using the spectrophotometer. The absorption peak in the visible range was found at about 525nm. The curve is quite linear in the concentration range of [0.005mM, 0.5mM]. This result indicates that KMnO4 could be a very good platform to quantitatively study nano-diffusion through our membranes, either on as-grown samples or on treated (like charge) samples. The only concern could be its stability – I will address this problem in the next post.
Below are the data table and the curve. Obviously at a concentration of 5mM the absorbance is no longer linear, which was not included in the curve plotting. But the linearity is very good within [0.005mM, 0.5mM] – our future experimental data should fall in this range.
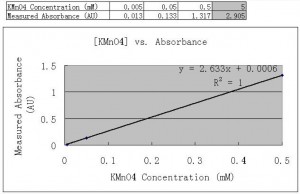
How’d you manage to get an R^2 value of 1? The spectrometer has an error reading of +/-.003 and after doing multiple experiments using the spectrometer the closest I got to a value of 1 was .998.
Also, absorbency values above 1 AU aren’t accurate, so they shouldn’t be used in the graph. When I made the concentration curves for Rhodamine I used multiple measurements and only used the data below 1AU.
I used Excel to plot the curve. I let Excel to insert and display a trendline. So the equation and R square are all generated values. Maybe they were automatically rounded to certain decimals.
The absorbance of [0.5mM] KMnO4 fits quite well. So I included it in the data. [5mM] is definitely not correct due to non-linear effect. We can try to use other concentrations like [0.1mM] to test its accuracy.