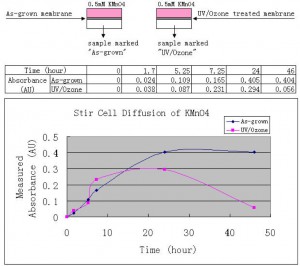
Stir Cell KMnO4 Diffusion Curves: strange and interesting
I got three pinhole-free membranes from Karl after the inspection. They are from wafer#612. Locations are (-3, 1), (-3, 2), and (-3, 3). I used the UV/Ozone machine to oxidize and charge two of them while leaving the third one as a control. The protocol of UV/Ozone treatment was followed.
The stir cell was set up as usual. The first three sampling was done within day one. The last two sampling was done after one and two days, respectively. The data and the curves are shown below.
When we designed this experiment, it was expected that the charged membrane would behave differently from the as-grown membrane. But the above curves show some strange behavior of the charged membrane. At first, I would attribute this to the stability of MnO4-, at least partly, if not all, given that MnO4- is a highly reactive oxidant. But it is still difficult to explain why the “UV/Ozone” curve goes back and forth compared to the “As-grown” curve.
We can notice that at the end of the experiment, both curves show certain trend of decrease. Probably because some of the diffused MnO4- got lost when they reacted with “something”. Interestingly, I found that both membranes were still intact after the experiment – anyway the highest concentration of KMnO4 used here is 0.5mM.
Was a buffer solution used? Is there a background salt concentration? What was the pH and was it constant throughout the experiment?
Reporting data in concentration would be more useful than AU, since you have data to do this conversion…
The KMnO4 was probably prepared directly without a buffer – I will check back. We didn’t measure the pH value of the solution before and during the experiment. Next time will do.
The data can definitely be converted to concentrations. Anyway it is linear dependence between AU and [C]. I will do the conversion and update the curves.