Various methods of applying PDMS to NC
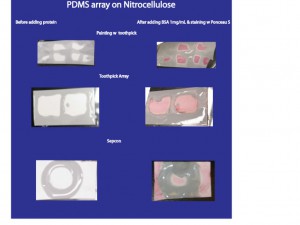
During the past week I tried various methods of applying PDMS to nitrocellulose. Below is a table of what the PDMS array looks like on the NC before curing in the over, and after applying 1mg/mL BSA and staining with Ponceau S.
When I painted the PDMS on, I used the thin side of the toothpick. This worked ok, but will not allow for uniform application of PDMS. I added 10uL of BSA to each “well” of the PDMS array and let sit for 1 hour. Then I washed the NC + protein with PBS for 30 seconds and covered with Ponceau S to stain for 2 hours.
Later I created an array using toothpicks, to stamp onto the NC after dipping in PDMS. This worked a little better, however, the PDMS spreads very quickly on the NC. Next I’ll try to add less PDMS to the stamp before applying it to the NC membrane.
I also dipped the top part of a sepcon into the PDMS and applied it to the NC. This worked pretty well, however the PDMS did spread during heating.
Chris mentioned the use of a heat stamp, however, I’m cautious to heat NC too much, because it’s highly flammable/explosive. However, I’ll read more about this week.
In later trials, when adding protein to the NC, I covered the entire membrane with BSA, including the areas covered by PDMS. I wanted to see if any of the BSA would leak through the PDMS. As apparent in the above illustrations, no BSA leaked through the PDMS onto the NC.

Do you think the PDMS spreads out more when heating? Does PDMS solidify in a reasonable period of time on the benchtop? Could we use a different temperature?
This seems to be in the right direction for an array. And the ponceau s staining looks great!
I notice that the PDMS begins spreading through the NC immediately up being placed on the membrane. However, it is quite possible that the heating causes it to spread even more. I’ll ask Henry about the possibility of turning the oven down. I’ll also try leaving the PDMS out overnight on NC, to see if it will solidify without the use of the oven.