Cell Adhesion Study (on silanized surface)
We have seen before that silanization slows down discoloration and hence prevents membrane degradation. In Sepcon, the sample should last over a week. Further, it was necessary to check whether silanized surface supports normal cell growth. Therefore, comparison of cell adhesion on aminosilanized surface is made to control surface (non silanized).
I took samples from W673 (no RTP) and amino-silanized following the usual protocol. Duplicates of both silanized as well as control samples were taken and assembled in Sepcon with ‘membrane side’ down. (NOTE: till now, all cell culture in Sepcon has been done with membrane side up; this was the first attempt to grow cells on ‘membrane side’ with this side facing down in a Sepcon.)
I sterilized the pncSi-transwell in 70% ethanol followed by rinsing in EGM (+FBS) and then conditioning in EGM(+FBS) in incubator before plating the cells on the membrane side. To plate the cells, I inverted the Sepcon so that the membrane faces up.
HUVEC (P4) were used at density close to 254,000 cells/ml. 10 ul of this was plated on each sample i.e. the plating density was: ~11700 cells/sq.cm.
After 4-5 hours, live adhered cells were stained using Calcein AM fro 30-45 minutes at RT. Five images were taken per sample and cells were counted using the MATLAB script. % Adhesion was determined with respect to the density of cells plated.
It was observed that,
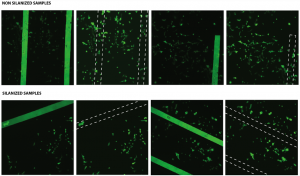
for non-silanized samples, % adhesion= 57.54%
and, for silanized samples, % adhesion= 52.71%.
The above values show that silanization doesn’t seem to effect the cell adhesion on pnc-Si. Further trials would confirm if there is any significant difference or not.
Following few images show the live stained cells:
As seen above, the morphology of the cells look similar in both cases. The white dashed lines show the slit positions and help to locate the live cells on the actual membrane.
Also, by plating the cells on ‘membrane side’ keeping this side facing down in a Sepcon transwell, makes imaging much easier. By growing a monolayer on this side, a differnt cell type can be plated onto the well side and can be seen through the slits.
Nice work, but the morphology doesn’t look very HUVEC like in either case … lots of spindle processes. I haven’t been impressed with the look of the cells in the last several posts on cell culture.
Can we do our best to look at surface contact angles? We need values before and after ‘incubation’ with FBS. If silane treatment is making the surfaces hydrophobic, it should impact cell adhesion and spreading, but it is likely that proteins coat the surface during spreading and perhaps they passify the surface and facilitate attachment just the same. Contact angles and adhesion values for non-silanized, silanized, and FBS-treated membranes would tell the story.
Also – What do we know about the difference in membrane stability when right-side-up or up-side-down membranes are used in the transwell format?
I agree. I will check the surface contact angles.
The membranes were found to discolor faster in transwell format with membrane side up.
I agree that the HUVEC have been looking weird lately. Not sure what to blame this on.
Since the endothelial monolayer is the most important layer for our future studies, I think we should switch to pnc-Si membrane side down cell culture from now on. This post showed that microscopy is no problem in this format (at least with this objective).
I’ll start looking into TEER in this format later this week.