Calcein vs. CMFDA staining
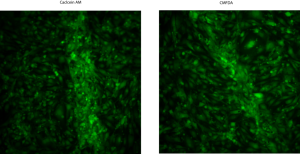
The other (see below) just-to-be-sure experiment I performed this week was to compare staining between 2 different live cell dyes. I typically use calcein AM to stain live cells green. This is a membrane-permeant dye which, once inside cells, is hydrolyzed by intracellular esterases to make it fluoresce green. Another very common live cell stain is called CMFDA. This dye is also membrane-permeant. After it enters cells, it gets thiolated (there are lots of thiol sources and appropriate enzymes inside cells) into a membrane-impermeant, green fluorescent compound. In this experiment, I wanted to confirm that black holes (vacuoles) in calcein-stained samples were due to some weird staining artifact.
These are P9 bEnd3 cells, seeded at 50,000 cells/cm2 onto SC501 transwells and allowed to grow for 1 day. I then stained 1 sample with the usual Live/Dead (calcein-based) protocol. For the other sample, I made 5uM CMFDA in media, stained cells in the incubator for 30 minutes, rinsed out the dye-supplemented media, incubated cells for another 30 minutes, rinsed in PBS and them imaged on the Nikon.
So, the lack of staining in cells over membranes is not due to a calcein protocol artifact.