Pore formation theory
In this post, I will present my thoughts on the possible mechanisms of pore formation based on our most recent observations with the substrate bias experiments. As a precursor, I will discuss silicon crystallization since it is intimately related to the pore formation process. I will try to show that pore nucleation is driven by stress induced by the bulk substrate as well as the point defect density and Si-SiO2 interface quality. Pore growth is explained in terms of Si atomic mobility and vacancy generation. I welcome any comments and areas of concern.
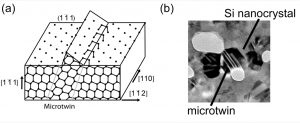
The solid phase crystallization (SPC) of amorphous silicon begins with the nucleation of crystallites followed by a subsequent growth stage. As deposited amorphous silicon is in a metastable state. It is modeled as a continuous random network (CRN) in which each atom has four other silicon atoms covalently bonded to it. Previous studies have shown that as-deposited amorphous silicon contains defects in the structure such as point defects and dangling bonds. These defects are sites for nanocrystal nucleation during a high temperature annealing process. Once a nanocrystal nucleates, growth proceeds in a step-like manner as depicted in the figure below. Growth is fastest in the <100> direction and slower in the <110> and <111> directions by a factor 2.5 and 25, respectively. The difference in growth rates is derived from the fact that only one Si atom is required to form the two bonds necessary for incorporation onto the (100) nanocrystal plane. In contrast, clusters of two Si atoms are required for growth on the (110) plane and three atom clusters for the (111) plane.
An important defect structure that results during crystallization is a microtwin. These features form at the amorphous-nanocrystalline interface on the (111) surface. Because three-atom clusters are required for growth on this plane, two bonding configurations are possible. Microtwins develop when a three-atom cluster forms on the (111) plane with a stacking order that is the reverse of the base nanocrystal. Growth along a microtwin ledge is also enhanced due to the decreased number of atoms (from three atoms to two) required to “join” the nanocrystal. Although no literature exists documenting void formation in silicon, voids have been observed near these defect structures in polycrystalline copper. Interestingly, pores are frequently accompanied by microtwin features in TEM micrographs of pnc-Si as well.
Nucleation of nanocrystals occurs mostly at the amorphous silicion-silicon dioxide interface. It is reasonable to assume that pore nucleation occurs at the interface shortly after nanocrystals begin to form. As we saw in my previous posts on the effect of substrate bias on morphology, application of a bias during the amorphous silicon deposition step greatly changes pnc-Si morphology. Without a self-bias, the a-Si/SiO2 film interface is rough with intermixed areas of silicon and silicon dioxide. This hinders nanocrystal nucleation at the interface and thus severely limits pore formation. Additionally, the presence of a dangling bonds in the bulk amorphous layer serve as nanocrystal nucleation sites which quickly grow into one another eliminating the possibility of pores to penetrate through the entire silicon layer.
One important consequence of a self-bias is that “high points” created by impinging Si atoms become mobile and they are able to rearrange into a lower free energy state. Prior studies have also shown that a substrate bias reduces the dangling bond density and oxygen contamination in an amorphous silicon film deposited by physical vapor deposition, thus yielding a higher quality material Thus, a substrate bias will create a smoother interface and reduce the number of defects in the bulk of the film.
We have seen that substrate bias produces a large non-linear response in pore size and density. We attribute the low pore density with no applied substrate bias to a rough Si-SiO2 interface and large defect density in the bulk of the film, which inhibits pore nucleation. The high defect density leads to Si nanocrystal nucleation throughout the film thickness, thus the likelihood of a void spanning the entire thickness of the film is greatly reduced as grains very rapidly grow into one another. A low substrate bias (2 W) reduces the bulk defects but still leaves a finite amount of point defects at the interface such that void nucleation can proceed. Higher biases (> 5 W – 50 W) produce low defect densities in both the bulk and interface regions. The reduction in point defects at the interface reduces void nucleation sites, which supports the observation of a sudden decrease in pore density at biases greater than 2 W. Interestingly, the largest pore sizes are seen at a 5 W bias and decrease with increasing bias. It is possible that higher power biasing over-densifies the amorphous silicon, which impedes the mobility of Si atoms and subsequently slows pore growth. At 50 W however, there is a significant increase in average pore diameter. This may be due to a finite amount of argon implantation into the silicon layer, which adds an additional growth mechanism. Interestingly, an increased density of microtwin defects has been observed in crystallized silicon films that were previously implanted with Ar. A higher population of microtwins would indicate that Si atoms would have an increased number of “paths” to move and create more vacancies, thus increasing the pore size.
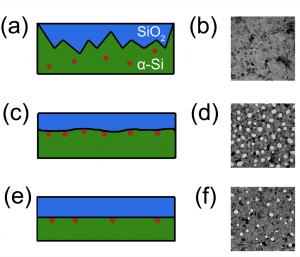
Figure above: Cartoon depiction of the SiO2-Si interface with different substrate bias powers during the silicon deposition and the associated TEM micrographs. (a) – (b) 0 W, (c) – (d) 5 W, and (e) – (f) 25 W. The red dots indicate the location of nanocrystal nucleation sites.
To date, there have been few instances in literature describing pore or void formation polycrystalline materials. In general, void formation is explained as a result of stress relaxation from a thermal annealing process in which point defects in a material migrate along twin boundaries to grain boundaries. Because prior work has focused mainly on thicker films (> 100 nm), multilayer grains form and the probability of observing a through pore is low. In the case of pnc-Si, if Si atoms are mobile enough during the crystallization and the grain size is constricted to a critical thickness, it is possible for voids to span the entire thickness of the film leading to a through pore.
The location of nanocrystal nucleation is critical to the pore formation process. If the majority of nucleation occurs in the bulk of the amorphous silicon, crystal grains will quickly grow into each other thus reducing the probability of a pore extending through the entire thickness of the film. On the other hand, if the majority of nanocrystal nucleation occurs at the amorphous silicon-silicon dioxide interface, void formation may initiate at the Si-SiO2 interface. A similar dewetting phenomena has been observed in silicon-on-insulator systems (as Joe presented at journal club). In the case of pnc-Si, the silicon layer is confined by two silicon dioxide interfaces. As Si atoms continue move to form nanocrystals, they leave more vacancies behind and eventually a void will span both Si-SiO2 interfaces creating a through pore. The nanocrystal-pore structure is maintained due to a reduction in the stress and free energy of the system.
Figure above: Simplified schematic of pore nucleation. The green and blue balls represent silicon and oxygen clusters, respectively. (a) Nanocrystal nucleation occurs at the Si/SiO2 interface. (b) As the nanocrystal grows in size, Si atoms leave vacancies that become voids. (c) Voids that penetrate the film thickness become pores.
Once a pore nucleates, growth proceeds in conjunction with Si nanocrystal formation. As the Si atoms move to join adjacent nanocrystals, they leave more vacancies behind thus enlarging a pore. Pore growth stops when all the amorphous silicon is converted to a nanocrystalline state. The size of a pore is ultimately determined by how mobile a Si atom is during the crystallization process. This is affected by the energy made available during the annealing step and the silicon thin film quality and composition. We know that pore size increases with higher annealing temperatures and longer annealing time due the increased energy made available for nanocrystal and pore growth. At lower temperatures, only a few small pores nucleate. This is because the Si atom mobility is restricted during low temperature anneals. It is also speculated that the concentration of amorphous material at the nanocrystalline silicon grain boundaries is higher for membranes with lower porosity. As more energy is made available at higher annealing temperatures, Si mobility increases and pores start to grow in size and the amorphous content is converted into nanocrystalline grains. The density of microtwins surrounding a pore will also affect growth, as these features increase the Si atom mobility during crystallization.
Figure above: TEM micrograph showing a pore at the amorphous-nanocrystalline interface. Pores nucleate and grow as atoms in the amorphous phase become mobile and are converted into nanocrystals.
There is a lot of work that needs to be done to show that this theory is correct. Based on conversations with Chris, here are my thoughts on how to proceed:
1) AFM work to look at the interface roughness of films deposited at various substrate biases. We need to verify that the substrate bias is indeed affecting the topology of the film.
2) Deposit a Si film with a gradient bias. That is, design an experiment where we tune the bias such that we can create an optimal condition at the Si-SiO2 interfaces and in the bulk.
3) Find a tool that will allow us to measure the defect density of an amorphous film. Conventional ways of doing this are electron paramagnetic resonance (EPR).
4) Measure the amorphous to nanocrystalline volume ratio. Ellipsometry can do this, but I’m not sure how sensitive it is. Based on the pore size changes we are looking at, it might be less than 1% change in volume.