Filtrate Concentration of the Barcikowski Particles Remain Discouragingly Low Despite Larger Sepcons and Higher Salt Concentrations
Because the Barcikowski lab intends to use the nanoparticles they sent us, relatively high yields are necessary for any step that cleans them up. This is unfortunate, because in every single separation that we’ve done (with one exception mentioned further on) we never see yields higher than about 5%.
We’d thought that the problem was the Debye length – because the samples we were sent came in very low salts (.6mM sodium phosphate buffer for samples 1-3, 0mM for sample 4 and the platinum sample) the electrostatic interactions play an important role. For instance, at .6mM NaH2PO4 (the lab didn’t specify if it was a mono-, di-, or tri-basic, so we use mono- as a worst case scenario in our calculations), the Debye length is 12.45nm. This means that for a 20nm Sepcon, each of the pores is essentially closed. Even for a Sepcon with 30nm pores, the effective pore size would be (30-(12.45 *2) = 5.1nm).
The problem, then, can be summarized in two separations. I prepared solutions of both platinum (a polydisperse mixture with particles between 4 and 40nm) and sample 1 (small peptides both bioconjugated to ~7nm gold nanoparticles and free in solution)at 5mM NaCl (sample 1 also has ~0.6mM NaH2PO4), added 400uL of each to two different 30nm cutoff Sepcons, and spun them for 5 minutes at 2000rpm. The concentration of sample 1 was less than 3%; the concentration of platinum was too small to be measured. At 5mM NaCl, the Debye length is about 4.3nm, meaning the effective pore size of the Sepcon should be 21.4nm. Since the size of the platinum varies from 5nm to 40nm and the size of the gold is supposed to be fairly consistently 7nm, something else is throwing a wrench at the bench.
Salt concentrations greater than 5mM NaCl are impractical, since, as shown below, the platinum particles aggregate at 10mM NaCl.
Here’s a size distribution by number (Malvern) of platinum at it’s normal 0mM salt concentration (peak 1 is at 20.6nm):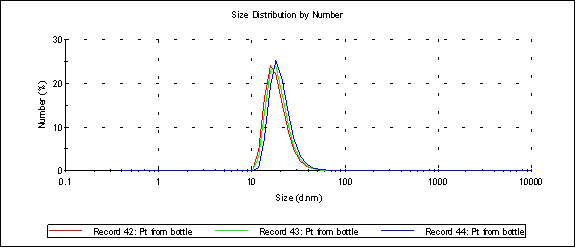
Here’s platinum at 1mM NaCl (peak 1 at 20.3nm):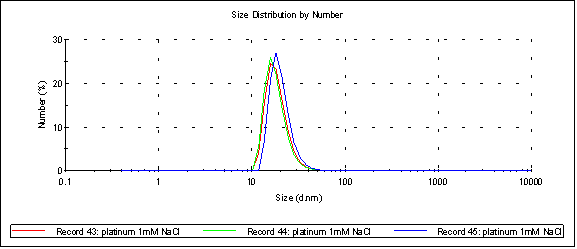
Here’s platinum at 5mM NaCl (peak 1 at 26.6nm, [red] at 7.97nm):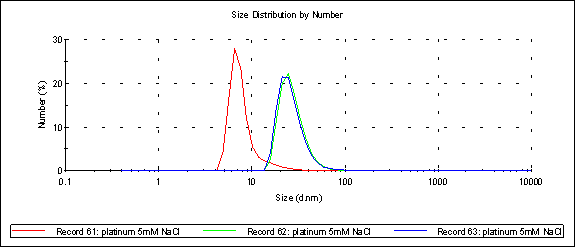
and finally platinum at 10mM NaCl (peak 1 at 110nm, although the first measurement [red] peaks at 44nm):
Also, I’ve run out of the platinum sample.
Two guesses: Either the samples really are cruddy after sitting out in transit, and most of the gold/platinum aggregates, or the gold/platinum is adsorbing to the Sepcons. The following Malvern distribution of unfiltered unsalted sample 1 seems to illustrate the aggregation of the particles pretty well: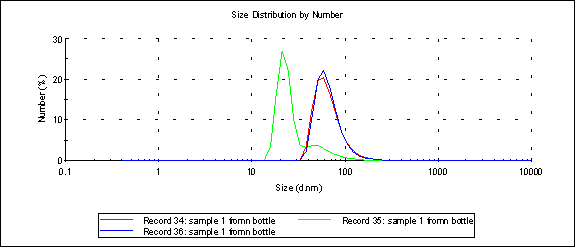 the first measurement [red] peaks at 21nm, but the overall size measurement is 67nm.
the first measurement [red] peaks at 21nm, but the overall size measurement is 67nm.
There are two (sort of) happy results – the best is that sample 3, which is gold nanoparticles without ligands, consistently makes it into the filtrate at concentrations of ~10%. Unfiltered sample 3 gives us a size distribution with a peak at 60.7nm:
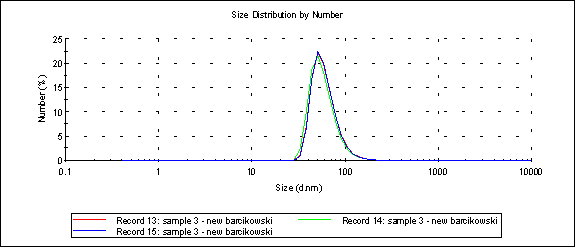 The Malvern gives the following size spectrum when the sample is passed through a 20nm cutoff Sepcon (peak 1 is 9.26nm):
The Malvern gives the following size spectrum when the sample is passed through a 20nm cutoff Sepcon (peak 1 is 9.26nm):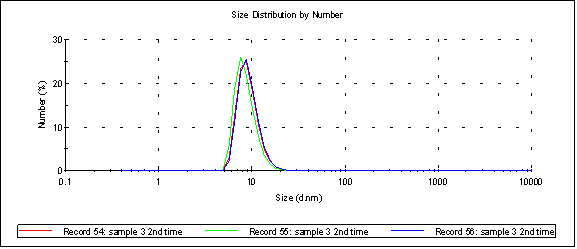
When the sample is passed through a 30nm cutoff Sepcon we get (peak 1 at 10.1nm):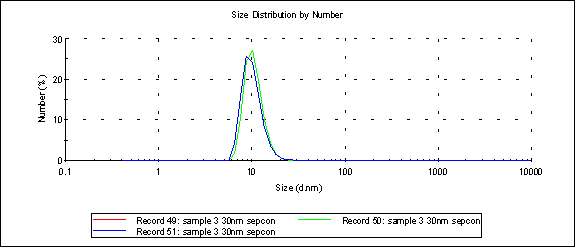
telling us that those particles are ~10nm in size, which is just a hair above the cutoff the lab wants.
Similarly, sample 1, when separated with a 10nm sepcon, consistently gives us tight distributions like the one below (peak 1 is 11.9nm):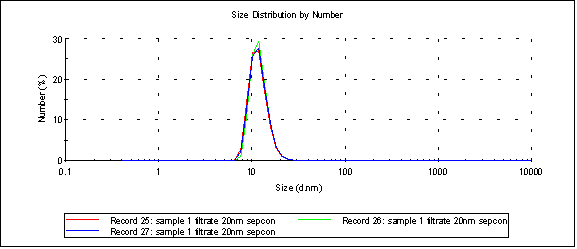
Which is great, but the concentration, as measured by the TECAN, is about 3% of the original sample.

1) Categorize your post
2) Please always include Malvern spectra so we can interpret raw data here
3) Did you run studies showing aggregation with the PT particles as we discussed. Were you able to explore the 1-10 mM range before running out of sample?
4) Can you explain the gold nanopartile separation that seems to be working? Is this a binary mixture or something more complex? Are we cutting the size after filtration? Again show spectra?