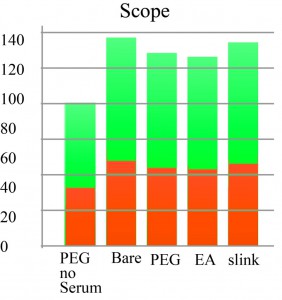
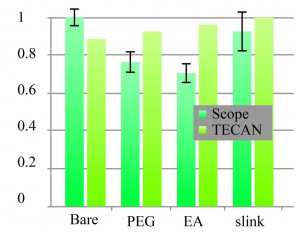
Serum adsorption, detection, TRITC, TECAN, Fluorecence 'Scope
These Adarza chips were pre wetted with PBS then incubated with fetal bovine serum (FBS) overnight in the refrigerator then rinsed. TRITC (2 mg/mL) added to chips and incubated at room temperature for one hour. Then rinsed once with reaction buffer then rinsed thoroughly with distilled de-ionized (DDI) water. I first measured using the TECAN (excitation wavelength 557 nm, whatever wavelength 576 nm with a gain of 40%) and then using the microscope. First thing to notice is that the scope and TECAN do not agree. Also, I am getting a higher signal with the scope than for the previous TRITC test. The first two graphs are the raw data. In the third chart, I subtract the value for the bare pnc-Si as background then normalize the two groups separately for their max valuse.
So if I disregard the TECAN, as it doesn’t agree with the scope, I’d say the PEG and EA reduce adsorption to only 70% to 75%. (25% to 30% reduction)

Thanks for the post. Here are some basic questions:
1) Is this fresh FBS? Commercially available and stabilized in some way? For this type of experiment, if any of the proteins have started to break down, they will likely adhere to almost any surface.
2) The overnight incubation – is there a shaker in the refrigerator or is this static? Static results will be very different than continuous flow dialysis.
3) What are the compositions and pHs of the various rinses and buffers that are used? You obviously don’t want to rinse or expose the chips to anything that will change the serum protein structures or destabilize them, and they will instantly adhere to surfaces.
4) Do the rinses contain any Tween-20 or other mild detergent? Without some type of mild detergent, even very weakly interacting proteins will not leave the surface. I think the primary concern is the strongly adhered molecules.
5) Why is there a DDI rinse? That’s the kiss of death for many serum proteins as they will unfold or denature. I guess it’s not a big deal as long as this is NOT the first solution used to rinse the chips after exposure. However, I still think DDI should be avoided in any experiment involving serum proteins.
I’m not sure that a reduction of only 30% is a result that I believe, but anything is possible. Experiments with actual membranes will make this more clear.
Regarding the next steps for the clearance experiments, I think you will need to go with the EA, since this is the only process that can coat samples with reasonable yield at this point. The PEG process still needs quite a bit of optimization since it breaks a lot of membranes, so unless you have a huge number of samples, it’s probably out of the question for Feb 5.
What the heck is ‘slink?’
Its hard to believe that serum is much different than FL-BSA. It might be good to have a FL-BSA control and do the TECAN study with FL-BSA to see if that squares with the scope.
I agree that we need to do clearance with EA because it is the only manufacturable coating so far. The stability of EA is a concern for a 30 hour clearance study, but we should give it a go anyway.
“slink” really should be “sLink” the coupling chemistry that Adarza has developed. As used in the post, this is basically a control sample of the amine-reactive surface that was not exposed to amino-PEG or to ethanolamine. It decomposes over time to a carboxylate decorated surface.
The EA will be stable, since it’s covalent, so should not come off. Whether it remains clean for 30 hours, remains to be seen, but probably will have a strong dependence on the experimental conditions and how stable the adsorbed serum proteins are maintained.
Thanks!