Single-Well CytoVus w/ SiO2 Membrane – Comparisons
I’ve run a few comparative studies with the CytoVu devices that have SiO2 membranes in them. From a previous NRG discussion, here is the comparison of a single location with only the fluorescent images, and that same location but with phase included:
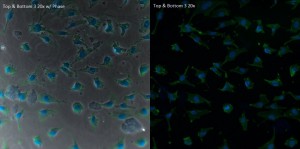
In the phase image, the pores can be seen through the cells, especially evident in locations such as the nucleus. However, the image without phase the pores are near-transparent and all but impossible to locate.
In addition, comparisons to various track-etched (TE) membranes was performed using the same protocol for fixing, permeabilizing, and staining. Four different TE membranes were used: two CoStar (one of 3µm pores and the other of 0.4µm pores) and two Greiner (both with 0.4µm pores, one considered translucent [low pore density] and the other transparent [high pore density]). For the CytoVu and all four TE membranes, cells were seeded on top (after a 1% Geltrex coating) and imaged fluorescently through the membrane.
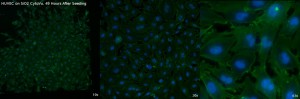
The first set of pictures is on a SiO2 membrane in a single-well CytoVu. The quality is good, with strong fluorescence of the cells and very little scattering or issues, even at 63x.
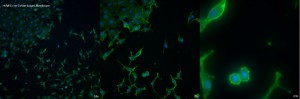
The first TE membrane to be looked at is the CoStar 0.4µm membrane. While the cells are visible well and there seems to be little direct interference, there are artifacts of the fluorescence visible especially at 10x and 20x.
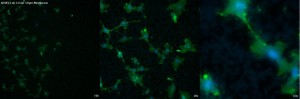
The next membrane, also from CoStar, is their 3.0µm design. In this membrane, the pores scatter the light to a much larger degree and cause warping and blurred appearance of the cells, in addition to the artifacts throughout the viewing area. While the nucleus and actin can be distinguished up to 63x, all three magnifications have low overall quality.
The first Greiner membrane (0.4µm Transparent) is similar to the CoStar 0.4µm membrane. While the cells are still mostly clear and bright, there are dots of light around signifying the presence of the pores. There also is more bleeding of the fluorescence than in other membranes, though this may have been caused by slightly different focal lengths.
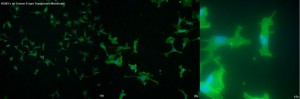
The final membrane to be tested was the Greiner 0.4µm Translucent membrane. Of the four TE membranes tested, this has the worst image quality. There is a blurriness to the cells instead of the crisp fluorescence desired. Also, artifacts which were seen in the other membranes are even more present and detracting from the cells in the image in this case.
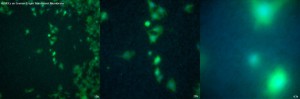
Overall, while not all of the TE membranes were significantly worse than the SiO2 ultra-thin membrane (the CoStar 0.4µm and Greiner 0.4µm Transparents membranes actually having rather good image quality), the oxide membrane has the best image quality.

The cells also look better spread on our stuff. Not surprising.
Can you summarize the NRG discussion yesterday?
The discussion was mainly just talking about the results and the SiO2 membrane’s optical superiority. The TE membranes apparently do look fairly good for how thick they are, but compared to SiMPore’s ultra-thin membranes they are still sub-optimal. There was some talk about the artifacts that appear in the dark space around the cells in the TE membranes; one suggestion was that the spots could be caused by dye trapped in the membrane. I ran a quick trial today looking at the membranes without any coating/dye/cells, and there are still artifacts. I’m guessing this is either some scattering and reflection of the signal, or perhaps some amount of auto-fluorescence of the membranes themselves.
Zach – are you coating the membranes in these pictures?
In reviewing this I’m struck by how much better spread the cells are on the SiO2.
Sorry for not getting back to you sooner, just noticed the question in the comment. I did coat the membranes in a 1% Geltrex non-gel coating before seeding with cells.