72 Hour Diffusion Test Results
I recently got some good looking results on the FLAG diffusion test over a 24 hour period. The relative concentrations of enzyme seemed to indicate that measurably more enzyme diffused from retentate to filtrate in solutions not containing constricting Anti-FLAG antibody, and this is excellent news, but over the 24 hours between beginning the diffusion and measuring none of the systems went to equilibrium. In the hopes of getting closer to equilibrium for more clear results, I ran another test over a 72 hour period.
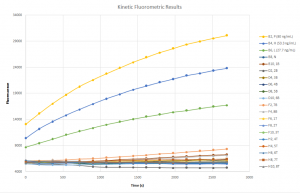
The top three lines here are three of my four standards, which were made fresh just before measurement. The lowest of the three (“L, 27.7 ng/mL) contains about a third of the enzyme that the highest concentration test case (sample 7) should theoretically contain at equilibrium, and other test cases should fall in around the area between the green and blue lines, samples H and L. However, all of the test cases were concentrated at the bottom. At first, I suspected cross-contamination — perhaps the samples had mixed due to poor bonding of PDMS to the new SiN material — but it seems that this was not the case.
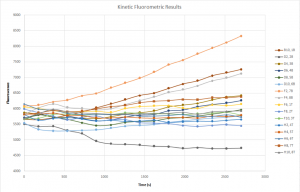
Upon further inspection, it’s clear that the general expected trends of the system were maintained. Forgive the extreme messiness of the plot — I’ll provide the salient interpretations. The sample I expected to be the highest, 7B, was indeed considerably larger than any other samples, and the negative control was the lowest. Other various expected trends held roughly true as well, and so I feel that the error lies elsewhere. Unfortunately, attempting to actually determine the concentrations of each of these samples proves tedious and unfruitful, probably due to the apples-to-oranges comparison that must be made between the test samples and the standard solutions. I suspect that due to adsorption and/or decay, much of the signal was lost in the test cases over the 72 hour period, whereas the freshly made standards haven’t had time to display such signal loss.
In order to remedy this, my first course of action will be to create the standards at the same time as the test samples. Any further difference in signal can be examined once the decay has been accounted for. I may end up storing my test samples in CytoVu assemblies sans-chips in order to better mimic the test conditions and get even more accurate results. In any event, all this will be picked up again in mid-January, when I return to campus. Happy holidays, everyone!