Kidney Week!
I went to Kidney Week in Philly, Nov 13-15. American Society of Nephrology.
I spent most of my time in the Bioengineering talks and posters. Here are some highlights.
I put the Abstracts pdf on the cloud.
Recellularization of Vascular and Tubular Compartments of Rat Kidney
Scaffolds with Embryonic Stem Cells
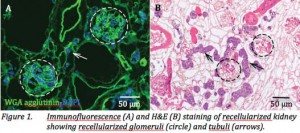
Basically, they perfused from the glomerulus and from the collection tubes. Using both together gave best results, but not all the scaffold was recellularized.
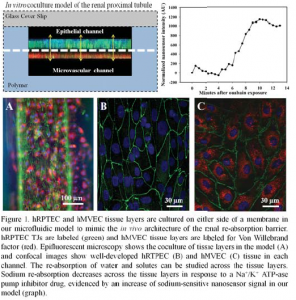
A Microphysiological 3D Model of the Renal Proximal Tubule Demonstrates Reabsorptive Function
Frohlich, Charest
Background: Re-absorption occurs in the proximal tubule as solutes and water are transported from the tubule lumen to the blood stream. Since the re-absorption of fluids and solutes in the kidney constitutes transport through epithelial and vascular components, models of reabsorptive barriers, such as the proximal tubule, should provide both phenotypes to more accurately replicate the physiological barrier architecture.
Methods: In this work we demonstrate a co-culture orientation of primary human renal proximal tubule epithelial cells (hRPTEC) and human microvascular endothelial cells (hMVEC) in a microfl uidic model. hRPTEC tissue layers were characterized by immunofluorescent protein labeling, gene expression, and barrier function quantification techniques. Re-absorptive function was investigated by quantifying molecular uptake and sodium transport across the tissue.
Results: hRPTEC tissue layers were found to have enhanced tissue structure, extended viability, higher proliferation rates, and increased levels of mitochondrial activity in coculture conditions of the model. hRPTEC tissue also expressed transport proteins and barrier characteristics, demonstrated glucose uptake, and showed decreased sodium re-absorption in response to a drug.
Conclusions: The physiological microenvironment of our model enhanced tissue structure formation in vitro and mimicked in vivo architecture and function of the proximal tubule. Our model allows observation of tissue structure development, direct quantification of kidney-specifi c transport mechanisms and a method to quantify functional change to kidney tissue due to injury, kidney active compounds, or toxic insult.
Long-Term Water Transport and Barrier Function of Proximal Tubule Cells Cultured Under Apical Shear Flow Conditions
Paul R. Brakeman, Fissell, Roy
Background: Development of a bioartificial kidney (BAK) comprising a hemofilter and a proximal tubule cell bioreactor for treatment of chronic kidney disease requires long-term survival of proximal tubule cells under shear flow conditions. Previous work has only characterized transport properties of proximal tubules cells for 2-3 weeks in perfusion culture. We investigated fluid transport characteristics of proximal tubule cells in a shear flow bioreactor in long-term culture for >60 days.
Methods: LLC-PK1 cells were cultured on polycarbonate filter supports for 14 days and then exposed to apical shear stress. Cells were perfused with media containing urea (40 mg/dL) and creatinine (10 mg/dL) on the apical side and media alone was used on the basal side. The change in volume of the compartments and concentrations of sodium, potassium, phosphorus, urea and creatinine were measured.
Results: Water transport for proximal tubule cells under shear flow conditions increased significantly from 34+/-10 ul/cm2/day on day 7 of low shear fl ow (0.2 dyn/cm2) to 119+/-12 ul/cm2/day on day 63 (p=.002) with high shear fl ow (2 dyn/cm2) and was stable for 14 days at high shear fl ow from days 49 to 63. We characterized the barrier function of the cells by looking at the leakage of creatinine. On day 63, the leakage of creatinine was 0.012+/-0.009 mg/cm2/day and did not differ significantly from the leakage of creatinine on day 7.
Conclusions: Proximal tubule cells can maintain barrier function and water transport under shear fl ow conditions for >60 days. Water transport increased with increased shear flow and time in culture.
Apical Shear Stress Enhances Organic Cation Transport in hOCT2/
hMATE1 Transfected MDCK Cells
Jayagopal, Fissell, Roy
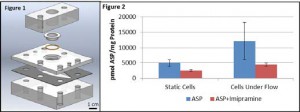
Proximal Tubule cells, snap well membrane. Measured electrolyte in apical well.
Sheer was ramped up over 1 month. Cells maintained under flow showed a 2.2 fold increase in protein concentration over static controls, confirming previous observations of shear stress effects. Furthermore, cells cultured under shear stress showed a 2.4 fold increase in ASP+ uptake when compared to cells cultured under static conditions and a 63.4±3.7% inhibition with imipramine compared to 48.6±5.5% inhibition in static cells
Conclusions: These results indicate that exposure to shear stress increases uptake of the active transport substrate ASP+ compared to static growth conditions.
Slit Nanotopography on Silicon Nanopore Membranes Resists Protein Deposition and Cell Attachment
Kim, Fissell, Roy
Surfaces modified by physically adsorbing either collagen type I (Col I-SNM) or covalently immobilizing RGD peptide (RGD-SNM).Atomic forcemicroscopy (AFM)was used to characterize the roughness of modified SNM surfaces.The propensity of protein adsorption on SNM surfaces was evaluated using fluorescein isothiocyanate labeled bovine serum albumin (FITC-BSA). Human umbilical vein endothelial cells growth on both modified and unmodified (Control) SNM were analyzed using immunohistochemistry.
Results: The surface roughness (RMS) of RGD-SNM (12.5nm) wasgreater than that of Col I-SNM (7.8 nm)and Control(6 nm). In unpatterned regions, FITC-BSA adsorbed strongly to the Col I as well as RGD, and RGD-SNM was found to significantly enhance cell growth (1500 % on day 7) compared to Col I-SNM (120%) and Control (100 %). In patterned area of all modified SNMs, however, FITC-BSA protein adsorption and cell growth are strongly attenuated (below 10 % on day 7). In addition, significant actin impairmentand cell detachment were observed on the patterned regions.
Conclusions: These results suggest that RGD is superior to Col I coatings for cell attachment. However, protein deposition and cell attachment on the slit-array region was significantly attenuated despite favorable coatings. This work will inform the development of SNM-based hemofiltration unit.
Nine-Day Filtration by an Implantable Hemofilter
Kensinger, Roy, Fissell
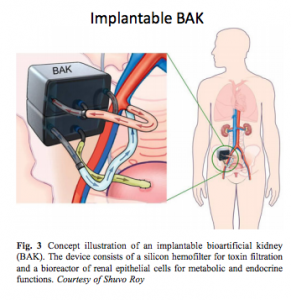

Methods: The device comprised a single channel blood conduit with two parallel-plate hemofiltration membranes (area per chip 0.7 cm2). Treated with biocompatible polymer – sulfobetaine methacrylate (SBMA). In vivo, the device blood conduit was connected via grafts to the common iliac artery and vein. 1.7 L/min. Filtrate was collected in two implanted reservoirs. The animal was housed without restriction and received thromboembolic prophylactic doses of enoxaparin (0.5mg/kg) once a day.
After 9 days, filtrate was sampled from the reservoirs. Albumin concentration in filtrate was measured by colorimetric assay. Filtration rates were estimated by indicator dilution.
Results: Effluent sampled on day 9 had low albumin sieving coefficients (bag 1: 0.13, and bag 2: 0.24). Ficoll sieving coefficients at equivalent Stokes radius were 0.14 and 0.19 respectively. In vivo hydraulic permeability was consistent with in vitro measurements after correction for plasma oncotic pressure.
Conclusions: A high efficiency hemofiltration device is capable of filtration over 9 days in a canine surgical model. Albumin sieving coefficients and hydraulic permeability in vivo match preoperative in vitro values. This demonstrates sustained in vivo filtration by silicon nanopore membranes without fouling or thrombosis.


These are slot channels, 7 nm wide, 100 nm spacing, the “active area” has 7% porosity.
Evaluation of Next-Generation Silicon Nanopore Membranes Optimized for Diffusive Clearance
Kim, Fissell, Roy
Background: Silicon nanopore membranes (HF-SNM) designed for hemofiltration have demonstrated remarkable permeability and selectivity. However, diffusive clearance was hindered by their thickness. Here we report hemodialysis-SNM (HD-SNM) with enhanced diffusive clearance.
400 nm thick membranes with 400 µm thick substrates. So they thinned the membranes at the active areas to 100 µm.
Methods: A new MEMS (microelectromechanical systems) fabrication protocol utilizing nested etch-back techniques was used to decrease the effective SNM thickness (HD-SNM 100 μm versus HF-SNM 400 μm). Diffusive clearances of polyethylene glycol coated (< 5 nm thick) HD-SNM and HF-SNM with sub-10 nm pore sizes were tested in a parallel plate flow cell.
PBS with Cr 10 mg/dL, BUN 90 mg/dL, and albumin 3 g/dL was recirculated (45ml), while dialysate (160 mEq NaCl) was recirculated in a counter-current fashion. At Qd=Qb=10 ml/min and zero transmembrane pressure (TMP) clearance was independent of flow rate. Solute clearance (K) was calculated by fitting concentrations measured at 0, 2, 4 hrs (n=3) to an exponential decay function: C(t)=Ci e-Kt/V. C(t): conc at time t ,Ci: initial conc, V: volume. Filtration was tested in water and fetal bovine serum at various TMP (1, 2, 4 psi) using cross flow velocities at 0.1, 0.5 and 3 ml/min. Platelet adhesion and activation were evaluated by immunohistochemistry (IHC) and scanning electron microscopy (SEM) after flowing human blood for 2 hrs at 2 ml/min.
Results: HD-SNM had a 3 fold improvement in K, consistent with mathematical models. Creatinine, BUN and phosphorus clearances were 280.8±20.8, 380.0±18.3, 231.1±7.6ml/min/m2 (HD-SNM) and 85.5±10.6, 135.3±22.9, 75.5±12.8 ml/min/m2 (HFSNM), respectively. HD-SNM maintained mechanical integrity at over 200mmHg. The HD-SNM also showed comparable filtration rates (71.5 ± 21.3 ml/hr/mmHg/m2) and selectivity to HF-SNM. IHC for CD62 and SEM images showed similar levels of platelet activation and adhesion.
Conclusions: These preliminary studies demonstrate signifi cant improvement in diffusive clearance with the HD-SNM while still maintaining mechanical robustness, selectivity, permeability and hemocompatibility.