Integrated Electroosmotic Nanopumps on a Chip
As part of my thesis work, I hope to create modular pumps and flow sensors using silicon nanomembrane technology. Devices that could generate/detect microliter and nanoliter scale flows would decrease the overall size of microsystems, replacing larger micropumps (pneumatic PDMS, Chip-sized peristaltic pumps) and costly detection schemes (laser reflectometry). These smaller devices would enable efficient scaling of ‘culture on a nanomembrane’ microsystems, as all of the necessary components to add the pump/sensing capabilities could be added with just a few additional metallization and insulation steps. In this post, I will briefly go over what has been done with our pnc-Si nanomembranes and what I hope to do.
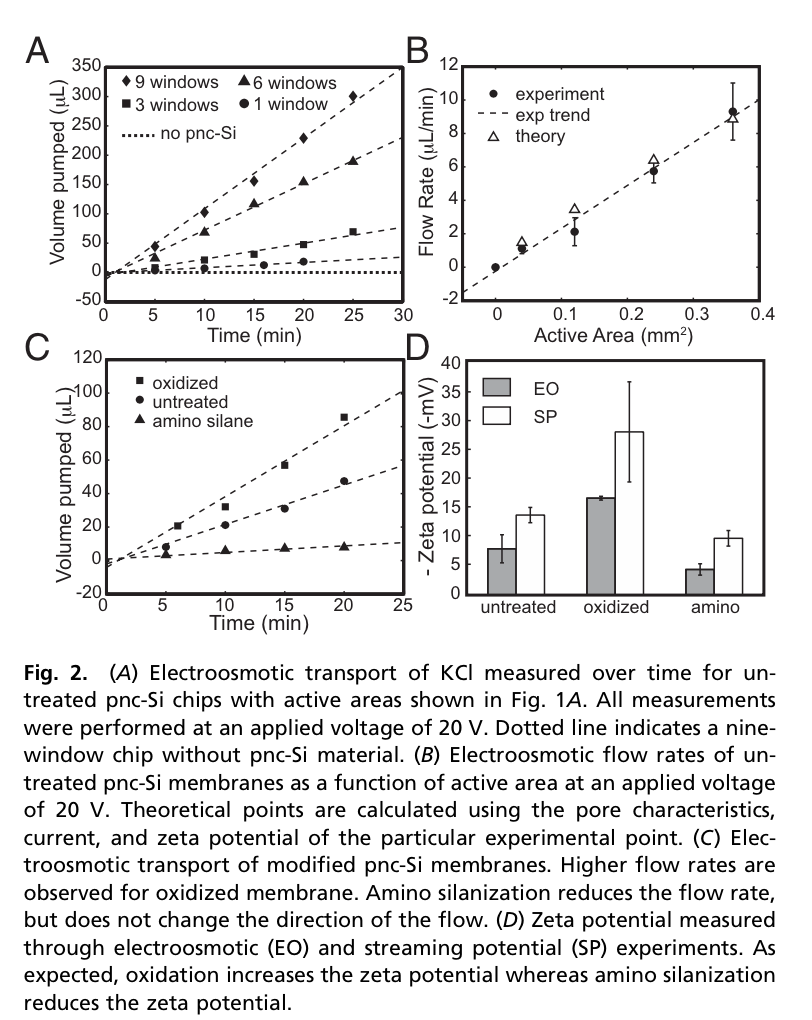
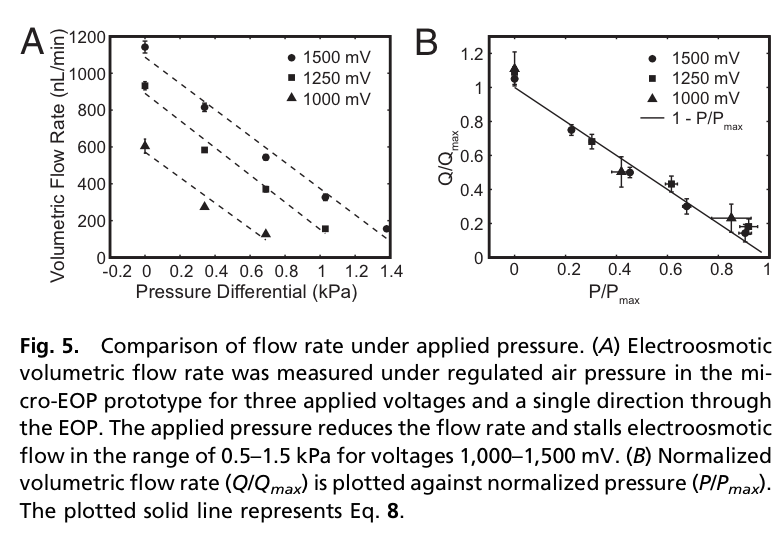
Jess Snyder’s PNAS paper (Snyder2013) outlined the basic behavior and theory for some small designed chips.
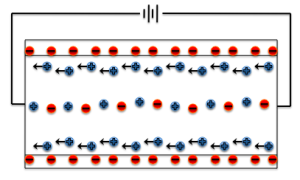
The major equations for electroosmotic flow in a single pore are:
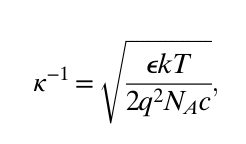
The key ideas are that the total amount of flow is affected by the capillary size and shape, the fluid involved in the motion (viscosity and electrical behavior), electric field through the pore, and the current through the pore. By summing up the flow contributions of individual nanopores, we can predict the total flow of many nanopores in the active area of a nanomembrane.
Advantages
- Directly scalable pumping behavior based on active area/pore characteristics of the pump. This means we can design chip layouts for a known voltage bias/material and expect proportional pumping.
- Tunable pumping behavior, based on the material of the nanomembrane and the applied bias. We can expect to modify the pumping rate by changing the applied voltage dynamically, which is fairly easy compared to some mechanical pumps.
- Low operating voltages. Everything could be powered by a AA battery in this system
- Easily reversed flow. Simply by switching the direction of the applied bias, the flow changes direction.
Disadvantages
- Requires reaction at the surface of the electrodes. Silver/silver chloride are generally used, as they are a non-polarizing electrode. However, this means that the electrodes are consumed eventually in the reaction (assuming solely pumping in one direction). Platinum could be used to provide hydrolysis, as a polarizing electrode, however, this leads to a lot of gas formation, which potentially could lead to bubbles. It is not impossible to remove some of the gas from the electrodes, or move the electrodes further away from the surface, but it is an additional consideration.
- Relatively low stall pressures (1 kPa). Could be improved somewhat by making the membranes thicker, increasing the hydraulic resistance of the membrane.
- Direct reactions with fluid inline, causing pH changes. This could be ameliorated by using a working fluid to run the pump.
- Thermodynamically inefficient, on the order of 0.0001%. The overall process is very inefficient, but we could improve upon Jess’ design by miniaturizing the system. The efficiency in an aqueous buffer was reported to be somewhere between 0.005% and 2% in Jess’ paper.
My initial vision for making the electroosmotically driven microsystem was motivated by a need for fluid flow in a cell culture system. I would manufacture a device using a combination of shadow mask evaporation and RIE to define electrode positions on a commercially available SEPCON chip, using gaskets sealed by pressure clamping or PDMS to define fluidic channels.
The video depicts a system where the electrodes are moved very close to the active areas of the nanomembranes, using a SEPCON chip as an example. Here, 2 membranes on each sided are used to push flow across (2x active area = 2x flow), with different voltages applied to each well. This allows for the net motion of fluid back and forth through the chip, based on the gasket design. The magnesium fluoride window would be used as a Raman observation space, though it could easily be any other silicon nanomembrane if Raman is not necessary. Depositing electrodes in all the wells helps to maintain the relative electrical behavior in all the wells, which is extremely important in electroosmosis.
I would change a few things about this particular design, shifting the electrodes further away, combining the active areas, and building a bubble traps near the electrodes. In spirit, this kind of device is something I believe we can achieve.