Forward vs Reverse Centrifugation (continued again)
As discussed in a previous post, we decided to test chips from wafer #1070 because they have smaller pores than those used in the most recent forward vs. reverse study.
In the forward direction, 31 uL of filtrate was passed after 30 minutes at 3 krpm. (after 30 additional minutes at 5 krpm the total filtrate volume was 65 uL)
In the reverse direction, 49 uL of filtrate was passed after 30 minutes at 3 krpm. (after 30 additional minutes at 5 krpm the total filtrate volume was 60 uL)
So it seems there is an advantage to the reverse orientation for the 1st 30 minutes at 3krpm, but that advantage is erased after 30 minutes at 5 krpm.
Below are the spectra for Absorbance and Fluorescence. They show that the forward orientation is not filtering the solution much as all, while the reverse is separating the 20 nm NPs from the IgG quite nicely.
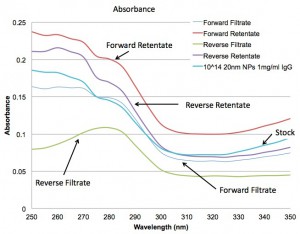
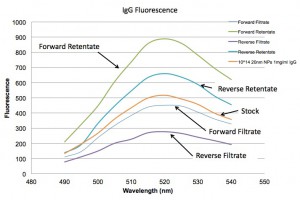
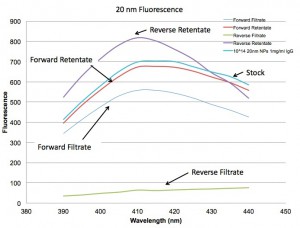
So are the assemblies leaking slowly???? I don’t think so because I’ve recently started doing work with the lipid micelles provided by the SJF group and they clog flow in the forward centrifuge such that there is no flow at all after 5-10 uL.
Jim suggested that I attempt the separation using constant pressure to see if we are still getting limited flow. I put the same solution from above into a 1085 filter and applied a constant pressure of 5 psi for 15 minutes and after the initial 5-10 uL there was no flow. I increased the pressure to 10 psi and the membrane broke. I don’t know why constant pressure would be clogging more quickly than a forward centrifuge setup.
As of now, the reverse centrifuge is the best way to get good separations using our membranes. The forward orientation in the centrifuge is generating more filtrate than I would have expected considering our results using constant pressure, but it also does not see to be exhibiting the same cutoff as we see using the reverse orientation.