ISEV-MRS 2019 Highlights
Hello Everyone,
Here is a summary of my trip to Nashville to learn about extracellular vesicles in cancer and present my work on isolation of nanoparticles/EVs using ultrathin nanomembranes.
Main points of the conference:
- This conference was about EVs in cancer, and did not include a lot about therapeutic applications of EVs. I could only follow the first 10 slides of each presentation talking about the background, isolation, and big objective of the project and after that i was almost lost since they were very deep in biology.
- Not a lot of talks about methods and technologies for EVs (including isolation).
- All the big EV companies were there including: Izon, NanoView, SBI, ONI, Beckman Coulter, Malvern, Spectrodyne, Nanostics, Codiak.
- Interestingly, somewhere between 60-70% of participants were non-americans, mostly europeans.
- In general, gave me a good picture of the most recent advances in the field.
- The struggle is real: everybody was trying hard to call the particles sEVs and not Exosomes.
- Everybody knows about problems of NTA and UC, however they still use them and they suggest EM for size characterization than NTA.
- Subpopulation of EVs is very hot topic in the field and this is could be our big contribution to the field.
- I also met with ExoDisc people (from Korea), i felt kinda attacked cause 5 of them showed on my poster and started asking questions. They are working on modification of their system to make it a single step (!!!!).
____________________________________________________________
Extracellular Vesicles in Cancer
Allissa Weaver (Vanderbilt University)
Link to the paper: http://jcb.rupress.org/content/214/2/197/tab-pdf
Main Points of the paper:
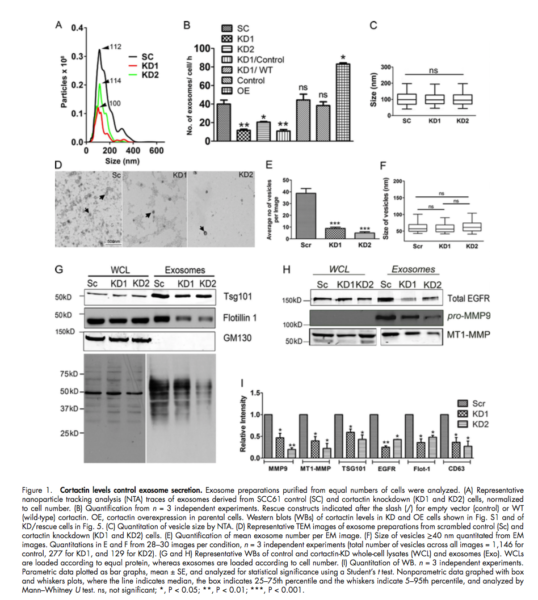
- Actin cytoskeleton regulatory protein cortactin promotes exosome secretion
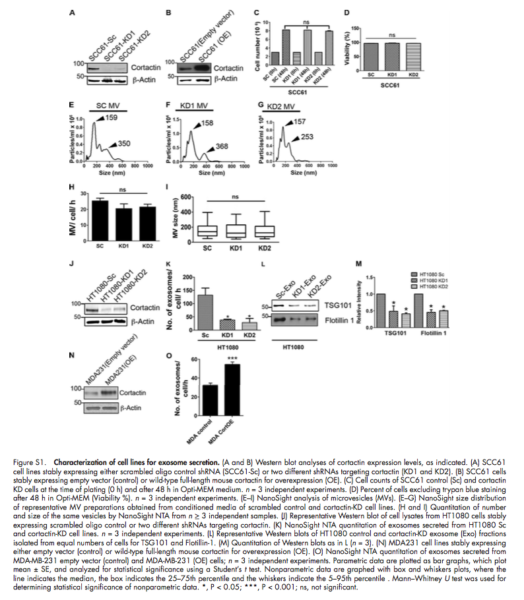
- Knockdown or over expression of cortactin in cancer cells leads to a respective decrease or increase in exosome secretion, without altering exosome cargo content
- Live-cell imaging revealed that coractin controls both trafficking and plasma membrane docking of multivesicular late endosome (MVEs)
- Coractin-knock down cell defects in serum-independent growth and invasion are rescued by the addition of purified exosomes using UC.
Experiments and Isolation:
- 10,000 g and 100,000 g fractions were compared for isolation of MVs and Exosomes.
- No difference in the mean number or size of MVs fraction (10000 g) between controls and cortactin knockdown EVs
- NTA data of exosome fraction (100000 g) revealed a decrease in the number of exosomes.
- The over-expression of exosomes was tested with TEM.
Background Information:
- Cortactin is an actin-binding protein that is overexpressed in many tumors and controls diverse branched actin-dependant processes, including cell motility, invasion and membrane trafficking.
- Cells were cultured in Opti-MEM which contains growth factors, so it sustains cell growth while avoiding contamination with exosomes from serum.
______________________________________________________
Biogenesis and functions of different EV subtypes in cancer-immune system cross-talk
Clotilde Thery
Link to the paper: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4776515/
Main Points of the paper:
- In the absence of demonstration of their intracellular origin, they should be called small extracellular vesicles (sEVs) because of co-isolation of mixed EV populations.
- Lack of purification and characterization tools prevents a clear understanding of the specific versus shared functional properties of the different EVs.
- They found several proteins often used in the literature as “exosome-markers” such as flotilin-1, heat-shock 70-KDa proteins, or major histocompatibility complex (MHC) class I and II proteins are present, to various extent, not only in the sEVs pelleted at high speed but also in larger/heavier EVs pelleted at medium or slow speed.
- Based on this analysis, we would like to propose the following categorization of EVs, which could be applied to any source of EVs, from cultured cells or from biological fluids: (i) large EVs pelleting at low speed, (ii) medium-sized EVs pelleting at in- termediate speed, and (iii) sEVs pelleting at high speed.
- Given the more restricted set of sEVs captured via CD63, and the general enrichment of CD63 in late endosomes, it is tempting to propose anti-CD63 as the preferred approach to isolate endosome-derived exosomes among the sEVs. Some cell types, however, secrete sEVs devoid of CD63, and for these it will be necessary to analyze instead either CD81- or CD9-bearing sEVs. In addition, given its presence in large EVs, CD63 cannot be used as specific exosome-isolation tool without a first step of elimination of these large EVs.
Experiments and Isolation:
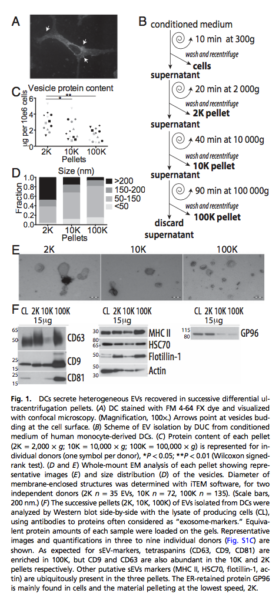
- Determining the specific markers of different subtype of EVs: Comparative analysis of the protein composition of all EVs recovered in different steps of differential ultracentrifugation protocol classically used to isolate sEVs.
- 24 hr conditioned media from human primary monocytes-derived dendritic cells.
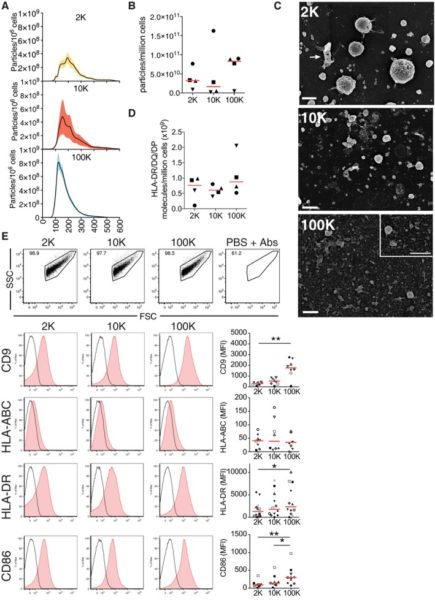
- Pelleted materials recovered by low (2000 g = 2K) or medium (10000 g = 10K) compared to ultracentrifugation speed (100000 g = 100K).
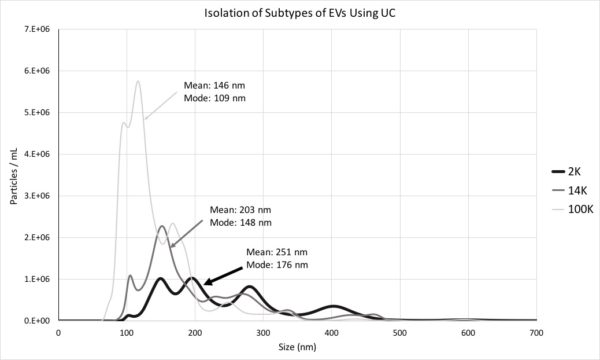
- 2K pellet: vesicles in majority larger than 150 nm, however, 50-150 nm vesicles were detected in 10K and 100K.
- Tetraspanins (CD9, CD63 and CD81) were highly abundant in the 100K pellet containing exosomes, but CD63 and CD9 were present, although to various degrees, in the 2K and 10K pellets.
- Floatation into iodixanol gradients allows separation of subtypes of EVs displaying different buoyant densities and sizes, with sEVs strongly enriched in the light fraction of the high-speed ultracentrifugation pellet (F3-100K).
Background Information:
- Density gradients, either of sucrose or iodixanol, are a classic means to separate membrane- enclosed vesicles according to their floatation speed and equilibrium density.
My conversation with Thery:
- She liked our idea of studying the effect of different size EVs derived from ADSCs on wound recovery of fibroblasts.
- She said the ultracentrifugation protocol needs to be optimized based on cell types.
- She suggested not to consider the non-EV associated fraction because of the low resolution of centrifuge.
They also published another paper in 2017 on subpopulations of EVs based on the pelleting properties of different size particles and they showed that for their application small EVs and Large EVs had the same effect. (This is very interesting for my experiments showing the effect of different size of EVs on wound recovery)
So based on this study, i tried to isolate different subpopulation of EVs using ultracentrifuge. I used 24 hours conditioned media of ADSCs (5 mL) and followed the following protocol:
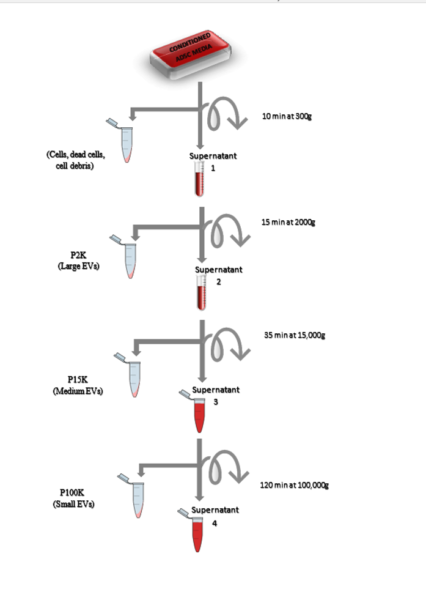
We named our samples P2 (Large EVs), P15 (Medium EVs) and P100 (Small EVs) and we checked the size of particles in each pellet using NTA:
________________________________________________________
Identification of distinct nanoparticles and subsets of extracellular vesicles by asymmetric-flow field-flow fractionation
David Lyden
Link to the paper: https://www.nature.com/articles/s41556-018-0040-4
Isolation Technique:
Link to the protocol: https://www.nature.com/articles/s41596-019-0126-x
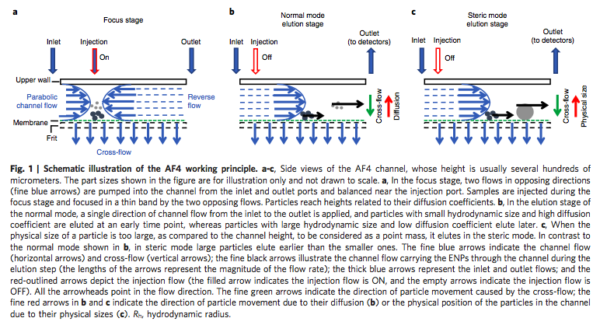
- Field Flow Fractionation (FFF): Developed by Giddings in 1966
- Separating analytes ranging from few nanometers to ~ 100 micron
- Device: A thin, flat channel (length of 10-40 cm and width of 1-5 cm) with a height varying between 50 – 500 micron
- Focusing step: To avoid spreading out particles in the buffer -> after injecting the sample, forward-flow and back-flow are applied
- An external field (perpendicular field) forces the particles and molecules toward the bottom channel wall
- The external physical field: Thermal gradient, magnetic, gravity or flow fluid
- In Asymmetric-flow field flow fractionation (AF4): the bottom channel is replaced with semi-permeable membranes with 10 kDa cut-off size
- The cross-flow is the driving force of accumulation of particles at the membrane
- The counteracting Brownian motion of the molecules results in their diffusion toward the channel’s interior
- The equilibrium of these two driving forces resolves the samples at different layers within the channel relative to the bottom wall
- A parabolic laminar channel flow that carries the samples forward from the inlet to the outlet
- Particles at higher positions will be eluted out of the channel as compared to less elevated particles
- Real time detectors for UV absorbance: Fractionated particles concentration
- Quasi-elastic (dynamic) light scattering: Particle size
Optimized parameters:
- Linear gradient of the cross-flow decreasing from 0.5 to 0 mL/min within 45 minutes
- Height channel of 490 micron
- Focusing time of 2 minutes with the same focusing flow rate as the injection flow rate (0.5 mL/min)
- Regenerated cellulose (RC) membranes better than polyethersulfone (PES): non-specific interaction
- Input sample ranging from 40 to 100 microgram
- It is important to note that samples were purified by ultracentrifuge before running with AF4
Like to a video: https://www.youtube.com/watch?v=4O3D2K28JbQ
Main Points of the paper:
- The purpose of this work; Developing the AF4 method and optimize the instrumental parameters to separate subpopulation of EVs
- The separation of samples is solely determined by the differences in their inherent diffusion coefficients
- Particles from 35 – 150 nm were successfully separated
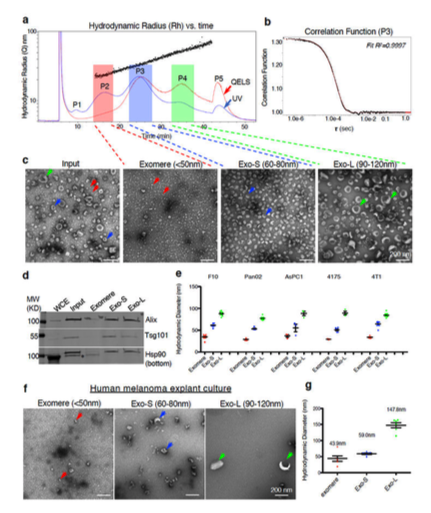
- Identification of five picks (P1-P5)
- P1: Represented void pick, a mixture of all types of nanoparticles
- P5: Composed of individual of aggregated particles/proteins (out of detection range) and eluted when cross-flow dropped to zero
P2: Exomeres
- Hydrodynamic diameter of 47 nm -> TEM showed a distinct population of particles not previously described smaller than 50 nm and lacked an external membrane structure
- Hsp90-b: highly present in Exomeres: Could be potential exosome marker
- Lipidomic analysis revealed that exomeres contain fewer lipid compared to others
P3: Small Exosomes (Exo-S)
- Hydrodynamic diameter of 62 nm -> TEM showed particles between 60-80 nm
- Enriched in proteins associated with endosomes, multivesicular bodies, vacuoles and phagocytic vesicles: Suggesting that Exo-S are most likely bona fide/canonical (derived from intraluminal vesicles of endosomal compartments)
- Flotilin 1 and 2: Specific markers
P4: Large Exosomes (Exo-L)
- Hydrodynamic diameter of 110 nm -> TEM showed particles between 90-120 nm
- Exo-L: Enriched in plasma membrane, cell-cell contact/junction, late-endosome and trans Golgi network proteins: May represent non-canonical exosomes or probably sEVs of different sub-cellular origin (plasma membrane budding)
- These 3 subpopulations were found in 20 different cell lines
- Almost all cells secreted higher amounts of exomeres relative to Exo-S/L
- Other previously reported exosome markers such as CD6, CD81, CD63, Tsg101 and Alix1 were present in both Exo-S and Exo-L