BSA-Au Separations
The BT method mentioned in this post seemed to give stable particles, but the BSA-Au did not work in separations. After about 24 hours, the particles retained pink color but seemed to settle in tubes. This may be due to reversable flocculation, not aggregation. The retentate and filtrates in separation experiments both lost all color, and by absorbance measurements seemed to have disappeared. This means the particles settled out or adsorbed to the walls of the tubes.
Previously I used another protocol that I got from a student in chemistry. I didn’t initially use this protocol because of the larger volumes of gold required. I went back to our old 5 and 10nm stocks (which are old and contaminated, but I have large volumes) to test out this old method again. Here’s the protocol:
1. Add 240uL of 1mg/mL BSA to 500 uL gold (higher concentration of BSA than BT method).
2. Aggitate for 30min.
3. Centrifuge for 30 min, 30k rpm.
4. Decant and resuspend in salt.
This worked well, but you can see here that the altered particle sizes by DLS are similar:
| Manufacturer | Unmodified | BSA-Au |
| 5nm | 6.8 | 13.1 |
| 10nm | 8.6 | 15.2 |
As can be predicted, there was no separation between the particles and neither particle went through the membrane:
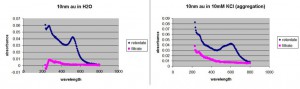
I wanted to see if the old 10nm particles could make it through the membrane in H2O and 10 mM KCl. It doesn’t look like they passed in water, and it appeared that the 10nm particles had a slight amount of aggregation by absorption: