MgF2 Cytocompatibility #4
Summary: I have some ok images (20x) of cells growing on MgF2. My controls still look poor. Something about the stain is causing the TCP + NPN controls to detach ahead of the MgF2 material, even if they aren’t fully dead yet. I used acetone fixation, which worked well for the materials on the nanomembrane, but attacked the TCP aggressively.


I performed another Live/Dead staining test with P5 HUVECs. They were seeded at 1e6 cells/mL on top of each membrane. Any spillover ended up in the bottom of the petri dishes, seeding the TCP.
Conditions:
2x 35 mm petri dishes with ozone bonded Chips
- Live Dish
- 3x 200um square window MgF2 (1 broke)
- 1x 700um square window MgF2
- 2x NPN
- 300 um gaskets with channels cut into them, exposing the TCP in the bottom of each channel
- Dead Dish
- 4x 200 um square window MgF2 (2 broke)
- 2x NPN
- 300 um gaskets with channels cut into them, exposing the TCP in the bottom of each channel
Procedure
- Seed cells, grow to mostly confluent (images taken at 24 hours)
- Image with phase only
- Apply Treatment
- Live – Rinse cells and stain
- Dead – Fix cells, Rinse, stain
- Image with Fluorescence + Phase
Protocol
For the fixation step, I used acetone. Acetone will permeabilize cell walls, so there was no need for Triton in this run.
Acetone Fixation
Add 3 mL ice cold acetone per dish.
Place at -20°C for 5 min.
Wash with PBS 3 times.
Results
Overall, the fixation did it’s job. Unfortunately, it also killed a lot of visibility on the TCP
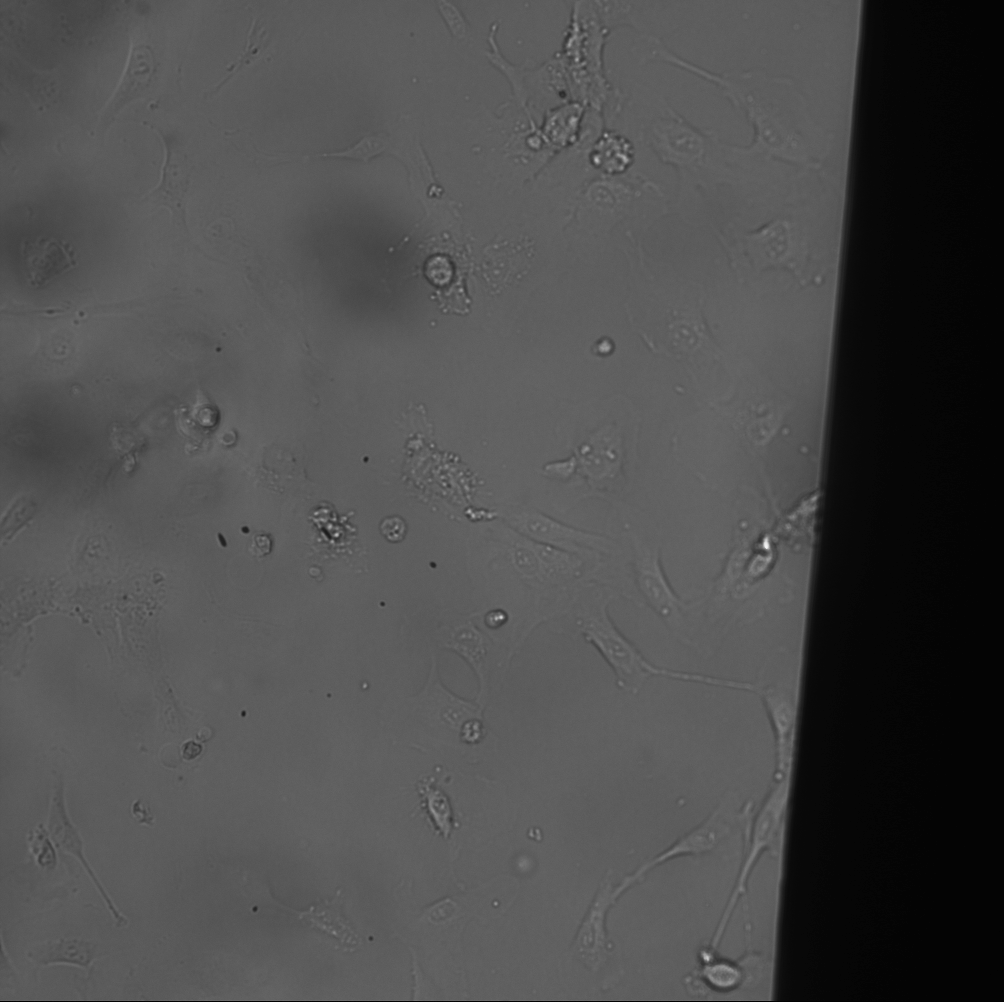
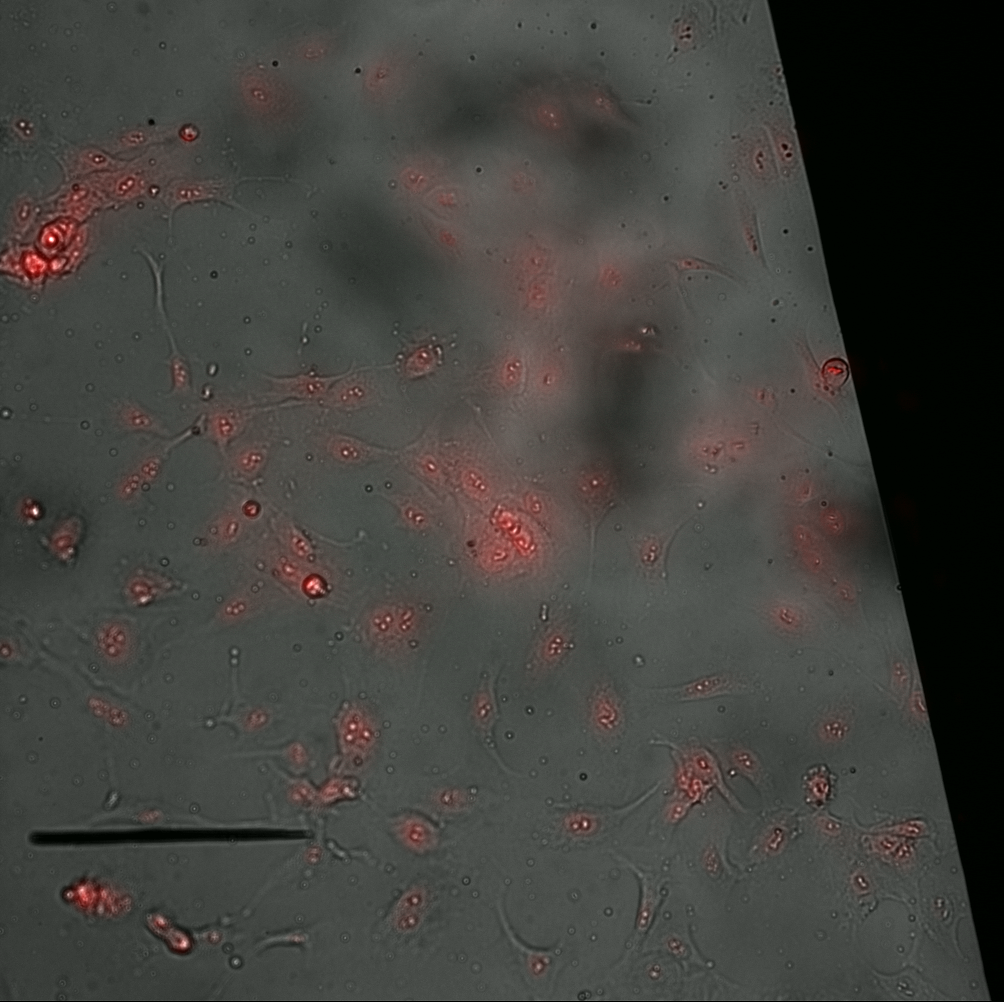
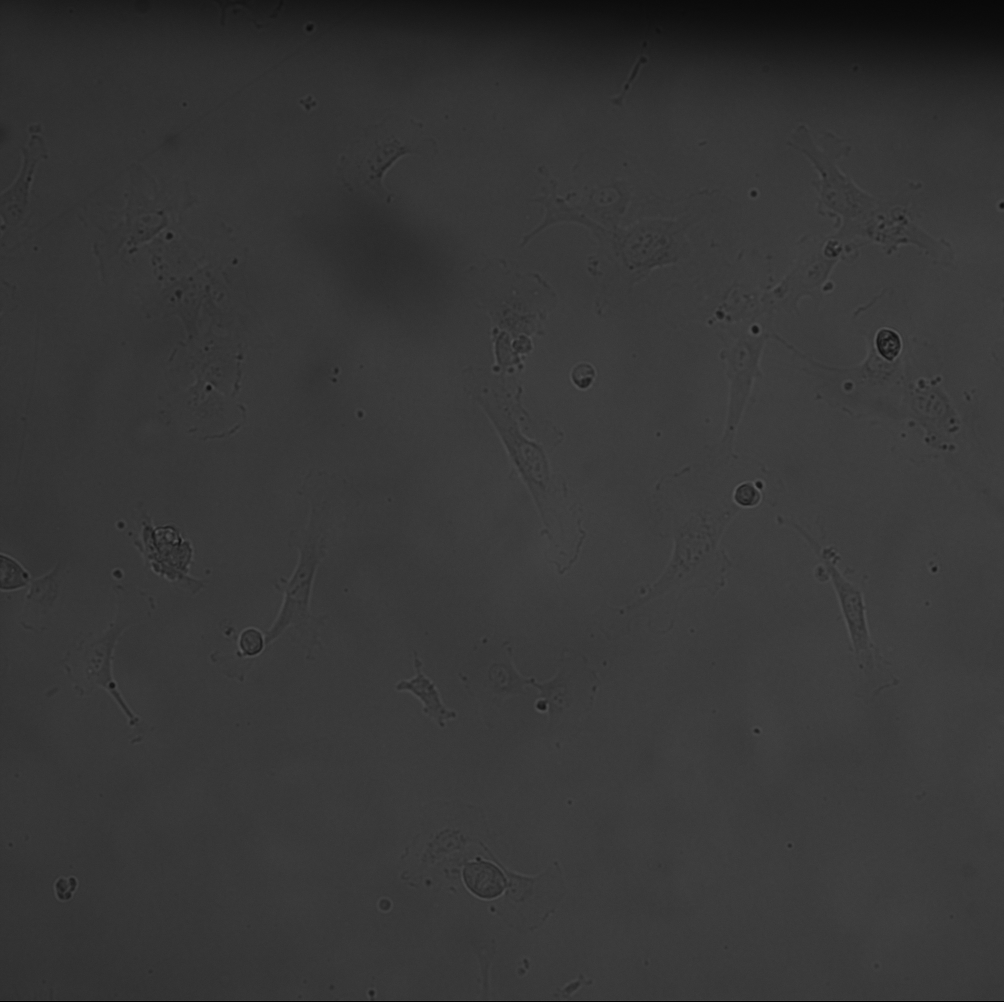
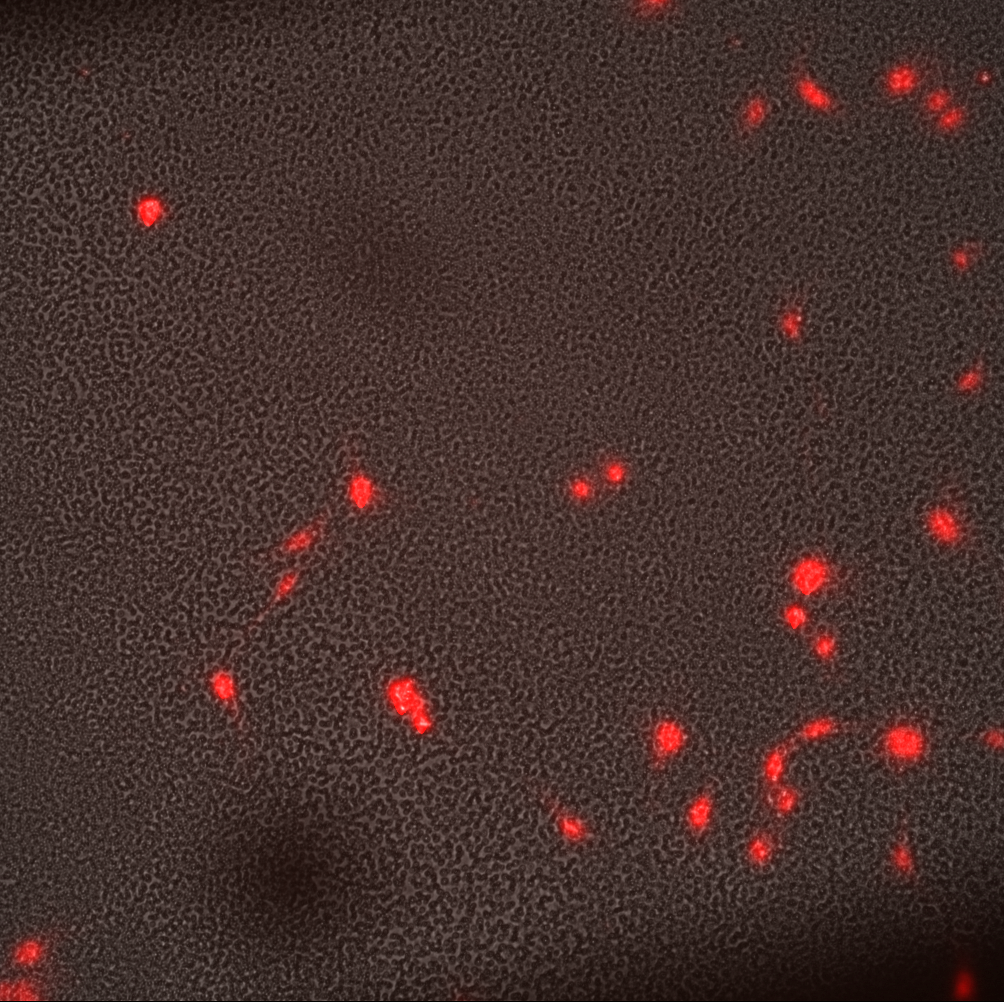
The MgF2 materials needed a larger window to see dead cells within the same window (see montage).

The cells began to ball up as time went on. It was particularly bad for the TCP and NPN. I imaged these treatments later compared to the MgF2, however, even then the MgF2 cells did not ball up at the same rate. I figure something in the stain or being out at room temperature for too long is causing this behavior. The imaging runs took about 20 minutes, after being incubated with the stain for 30 min.
I need to rerun a TCP control for sure. We are nearly there.