Possible sort of a partial success with NPN pre-filter
Since the control experiments seem to be going OK I decided to try again with one of the NPN-tented structures that Greg made originally. Since piranha cleaning seemed to be making a difference I wanted to pre-clean the chip, but from previous experience with a couple of poorly-tented NPN chips as guinea pigs, the NPN gets destroyed by Piranha in all but a few spots. The left images are the NPN before, the right ones NPN after the piranha treatment (orientation is preserved on top set, and it rotated 180 degrees on the bottom set) – these are not the chips I used for the experiment, to be clear, I just wanted to show you what piranha does to poorly tented NPN:
Note that there are still a few patches of surviving NPN, usually the best visual contact areas. So it seems that lamination strength is not uniform. Might be related to the surface of the substrate during testing.
So, while piranha is great before tenting, I need a more gentle clean for the pre-tented chip, so I used 2 minutes at 60 watts in air plasma. The images below are before and after the treatment, and the NPN is unaffected visually:
Note that this is one of the ones Greg made a long time ago using the dry transfer method (I think?) so it is not as flat as the newer ones and there are some channels.
Wetting was complicated by a leak in the cell, which necessitated removal and drying of the chip to fix. Another 1minute in the plasma oven to bond gaskets solved the issues, and the NPN was still visually intact, but could have been damaged a bit at this stage. Nevertheless, when I finally mounted and wetted the chip, the NPN was properly laminated.
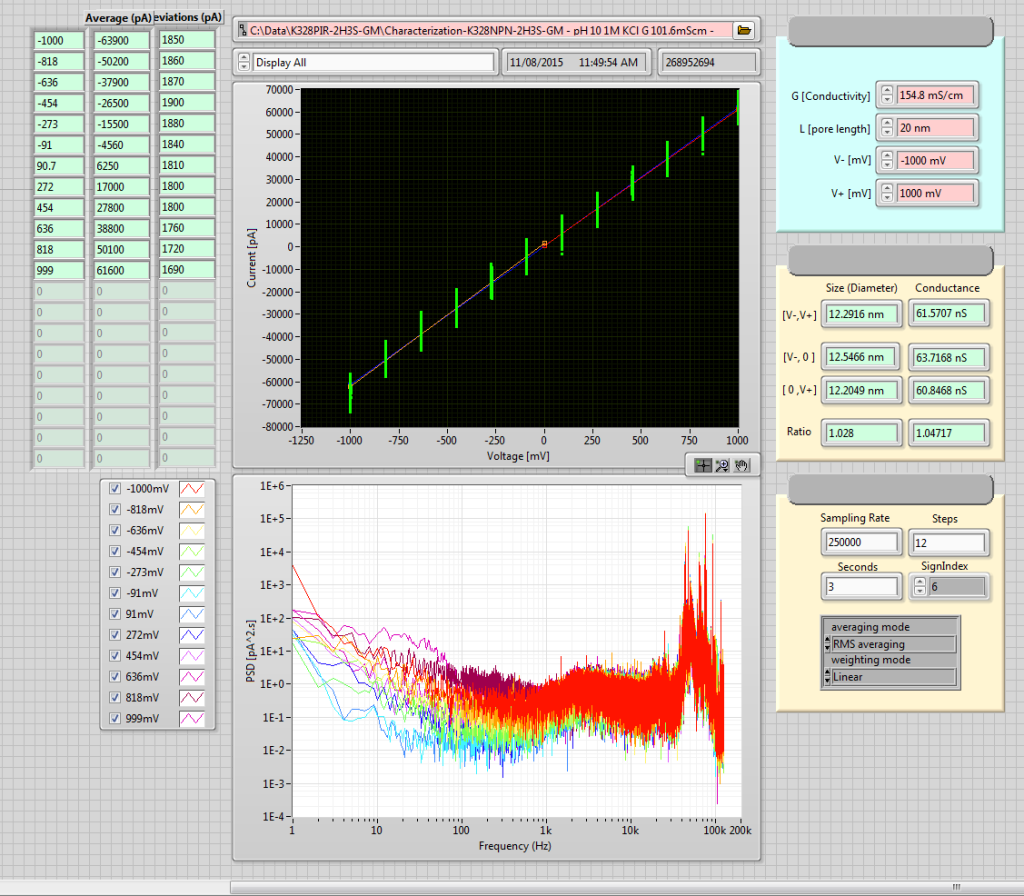
Fabrication of the pore was a little unusual in that there appeared to be something resembling a hole already in the chip. 
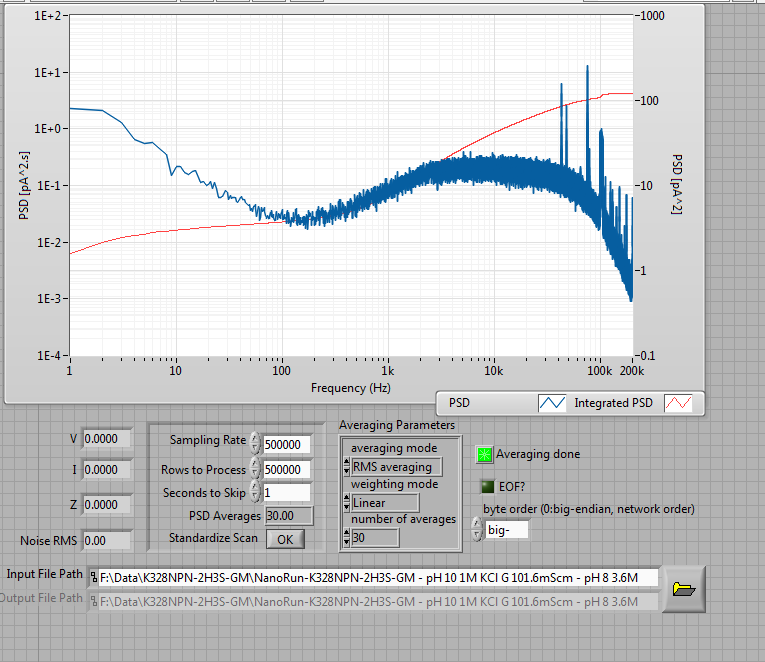
I went ahead anyway, and opened up a larger, more linear pore after about 2 minutes (fast for this voltage, indicative that there really was a defect there to begin with. Not necessarily a problem). The final pore was remarkably low noise and linear:
The baseline was clean before the addition of DNA
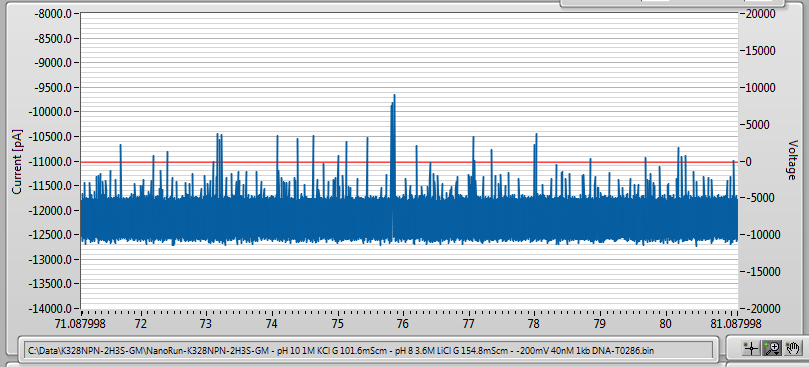
After adding 42nM of 1000 bp dsDNA, I saw this:
Finally, some results…
A did a couple of other voltages (-50, -100, -200, -300mV) quickly and then immediately dismounted it to see if the NPN had stayed on the chip. It had not, or at least not entirely:
You can see that while the NPN is still there, it is not in contact with the membrane so it is likely that the high event rate is due to DNA entering from the side.
This is disappointing but not entirely surprising give the high event rate. For such small NPN pores, I am assuming that they will present a pretty significant barrier to passage since they are not directly biased, so I would be surprised to see an event rate comparable to the bare case.
This suggests a couple of possibilities:
1 – it is possible that the original dry transfer method is just not as good, and not as stable as the new method which you guys have been testing for the last little while. If that’s the case, I just need to learn to use the new method properly (still have not tried, that’s next, but I’m getting pretty low on chips).
2 – the CBD method itself is causing delamination. We have a handful of experiment under optical microscopes showing that bubbles occasionally (but not usually) form during the high voltage step, likely due to electrolysis of the water. It’s possible that we are driving much smaller bubble formation during the high voltage step which are big enough to lift off the NPN.
Unfortunately I don’t have an intermediate image of the membrane to know if the NPN was still tented after pore fabrication and before addition of the DNA, but it is unlikely. So my next steps are:
1 – repeat experiment with pre-tented chip, but look to see if NPN survives the fabrication step. If not…
2 – make a pore in an untented chip, dry it off, clean it in Piranha, then tent the NPN on it and try to get DNA through it. A pain in the neck, but if it is the high voltage step causing stability problems this should solve it.
I will do a few practice runs with the new tenting method on some other useless chips, since I have not yet had a transfer nearly as nice as the ones I have been seeing in blog posts from you guys recently. I think I am going to just try to use my breath – the atomizers I got all have droplets that are far too big, and since this thing is a filter, it should at least keep any unwanted stuff in my breath away from the pore, right?








Getting closer. It looks like the NPN is still there but that the wrinkles grew into large pockets. The vapor transfer is much better at eliminating initial wrinkles and so hopefully that method leads to success. Yes the membrane is a filter and should keep all the crap away from the nanopore, but it is a shame that we haven’t found a substitute for this very crude method. Let me double check with the team to see if this is still the best we can do here.
I know you are low on oxide chips and Vincent is ordering more soon. Are you also low on NPN chips?
I have more NPN chips than I do oxide chips at the moment. I can probably also clean the NPN off some of the ones that came pre-tented that did not work too well using piranha and try to re-apply.
But it’s a good thing that new chips are being ordered.
In other news, it is surprisingly difficult to find conditions in which polystyrene nanoparticles will go through nanopores. I’m not sure if it’;s because the resuspension is not working or if they are just too fast to see, but I’m going to go back to DNA for the moment and try to get an NPN ship working with simple DNA.
I’m surprised that you’re having trouble getting nanoparticles through. What dimensions of pores and particles are we talking about here? It’s possible for particles which are technically smaller than the pores to still have trouble translocating — as a rule of thumb, you’d want particles a third the diameter of the average pore or less before you’d see unhindered transport.
While we haven’t done any experiments that I’m personally aware of on the transport of very long molecules like DNA through our nanopores, roughly spherical particles like globular proteins typically pass through our nanomembranes almost as if the membrane wasn’t even there. You shouldn’t expect to see a very large hinderance of transport of spherical molecules to your single nanopore with the addition of the nanomembrane filter unless there are other factors at play.
I was using 40nm pores for 26nm particles, so that could be the problem. Do you have a reference for that that I could look at in more detail? Or are you referring to diffusive transport when you say the pore should be 3 times larger than the particle?
Since we are using biased transport I would be surprised if you needed a 3x larger pore for transport, since we get microns-long DNA through nm-sized holes without issue – but that’s entirely due to the biasing, diffusion would never get anything through.
It could be that our conditions are just not right and the particles are not getting resuspended properly, but we are working in a regime where other groups have done it and the manufacturer suggests should be fine (10-100nM salt, pH 7-8).
Just ran another experiment on the same sort of chip – one I transferred with steam this time, but still fairly wrinkly. I took it apart after fabrication, after conditioning, and again after running the experiment. The delamination seems to happen gradually across all of those steps, without any obvious correlation to any of them. Seems like it’s just bad transfer that does it.
This one looks a little more promising in that it’s not immediately obvious that there is a fluidic path connecting the membrane area to the bulk solution, but it’s wrinkly enough that we can’t rule it out.
So the next plan is to piranha clean some chips, leave them overnight to get a little hydrophobic again, then tent NPN using breath and see what happens when the transfer is smoother.
Sorry for the late response. Nice work. Your inconsistent transfer looks like something that Tucker and I figured out during the breath vapor transfer process: the angle at which you hold the substrate to the vapor matters in getting even wetting and delamination. Doing it 0 degrees to the normal resulted in images much like what you are seeing. If I held the substrate at a glancing angle (45 degrees or more), it seemed to even out the islands of liquids.
If you are already doing angled vapor tent transfer, you could try and nucleate the transfer process by breaking the membrane at a corner, but this seemed to be irrelevant after figuring out the angled vapor transfer.
Yea these are the old ones, figured I might as well use them. I am going to try the angled transfer today, I’ll let you know how it goes.
I tried 5 transfers with a few tweaks each time, and got two of them working. The first three I tried just using breath and then either simply lifting off the chip, or breath then poking out the corners and lifting off – no luck. Wrinkly and poorly adhered so not all the membrane stayed behind.
The last two I used breath to laminate and then steam to adhere before removing from the jig and those both worked well (at least they look good, we will know for sure once they are wetted.
The three failed ones can go back into the Piranha bath to remove the NPN so I can reuse those chips as well.
Progress!