Investigating the endothelial basement membrane formation on SiO2 membranes
In our prior work we showed that endothelial cells form far fewer focal adhesions on microporous SiO2 membranes compared to non-porous SiO2 and tissue culture plastic (see prior post by Ana). We performed this analysis at 24 hours, which seems to be the standard in the literature.
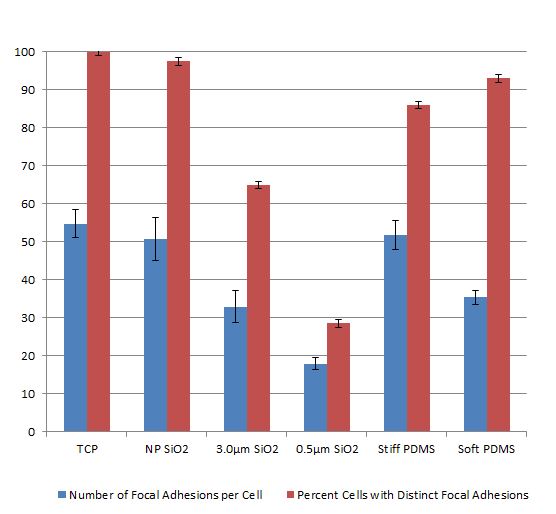
Interestingly, we found that the number of FAs over the microporous membranes substantially increased by 6 days. We also found that the endothelial cells formed mature barriers by the presence of junctional proteins (VE-cadherin) regardless of substrate porosity or pore size. This suggests that while the substrate porosity and contact area dictates early cell-substrate interactions, it does not affect barrier maturity as long as the cells are able to hang on and proliferate.
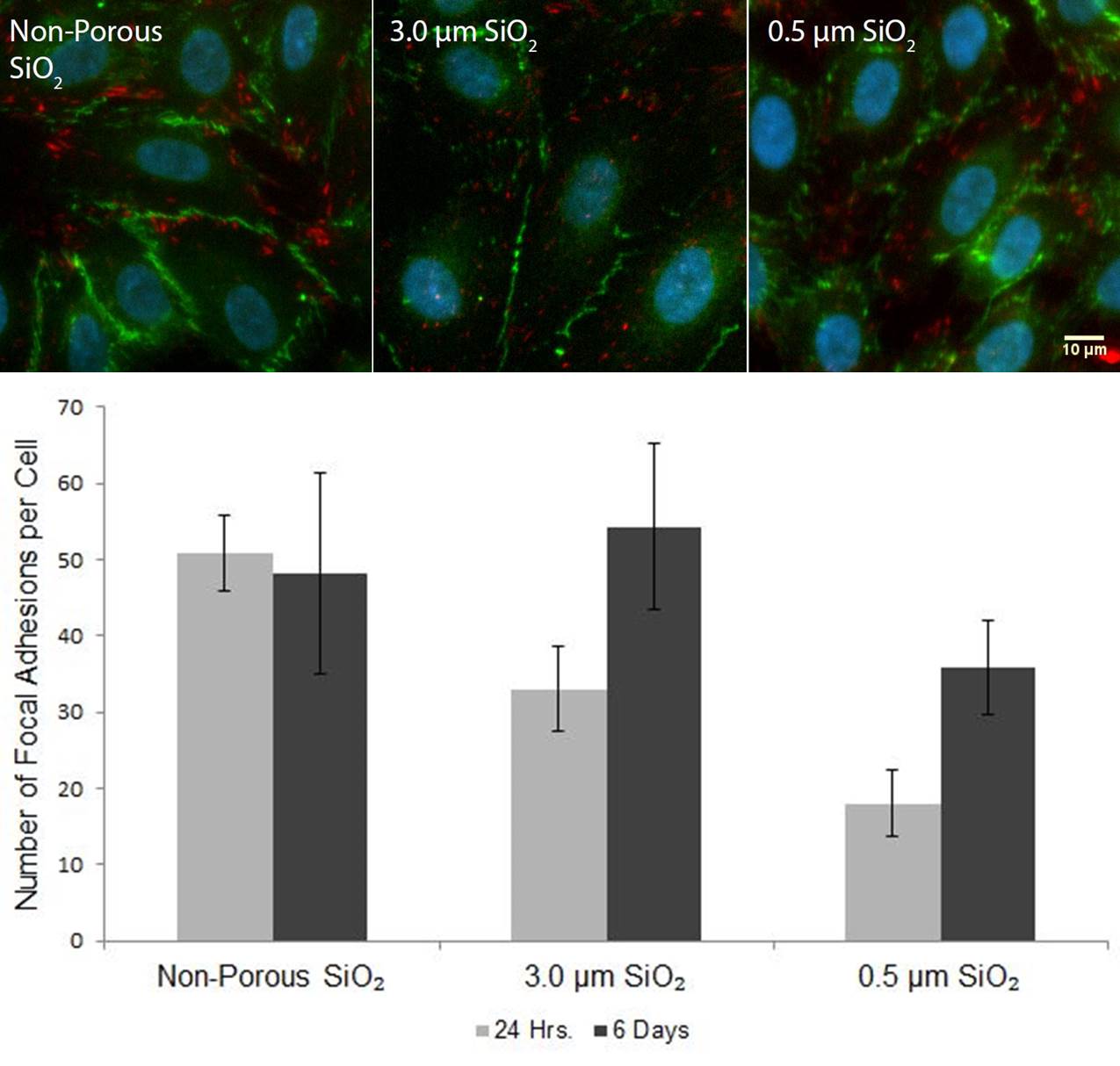
We hypothesized endothelial cells were able to form additional focal adhesions due to their deposition of a basement membrane or extracellular matrix over the open pores. This was inspired by the observation of FAs over pores at 6 days in overlayed fluorescence/phase images.

High-resolution fluorescence image of focal adhesion formation on 3.0 µm pore size SiO2 membranes upon reaching confluence. Cells were stained with DAPI (nuclei, blue), anti-VE-cadherin (cell-cell junctions, green), and anti-vinculin (focal adhesions, red). White circles are locations of pores. Solid circles are locations where focal adhesions are present over the pore or at the pore edge. Bar = 10µm.
Since cells attach to basement membrane proteins fibronectin and collagen IV via focal adhesions, we set out to fluorescently stain for these proteins on 3.0 micron high porosity membranes and TCP controls. We found minimal staining of collagen IV at 24 hours. In several places there appeared to be dark holes in the location of the micropores. Staining was also minimal on TCP.
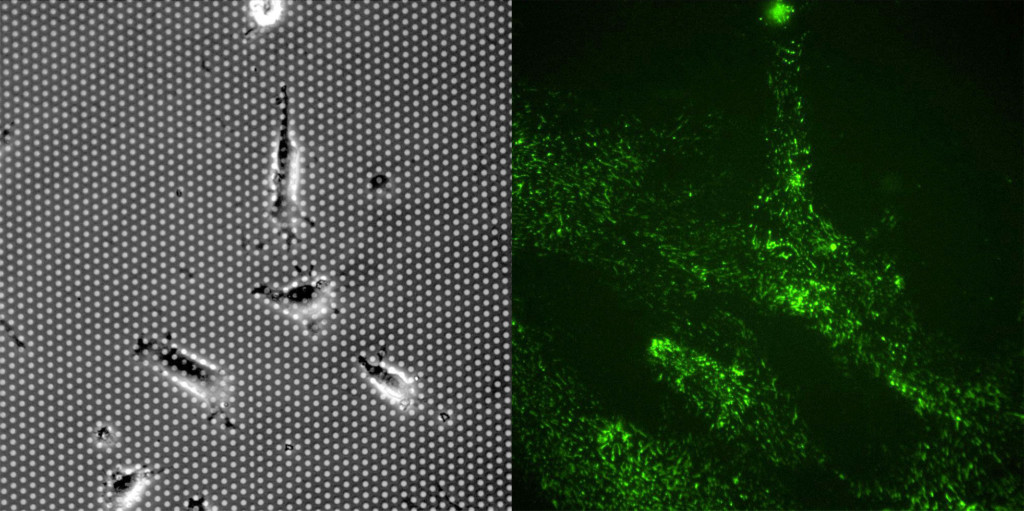
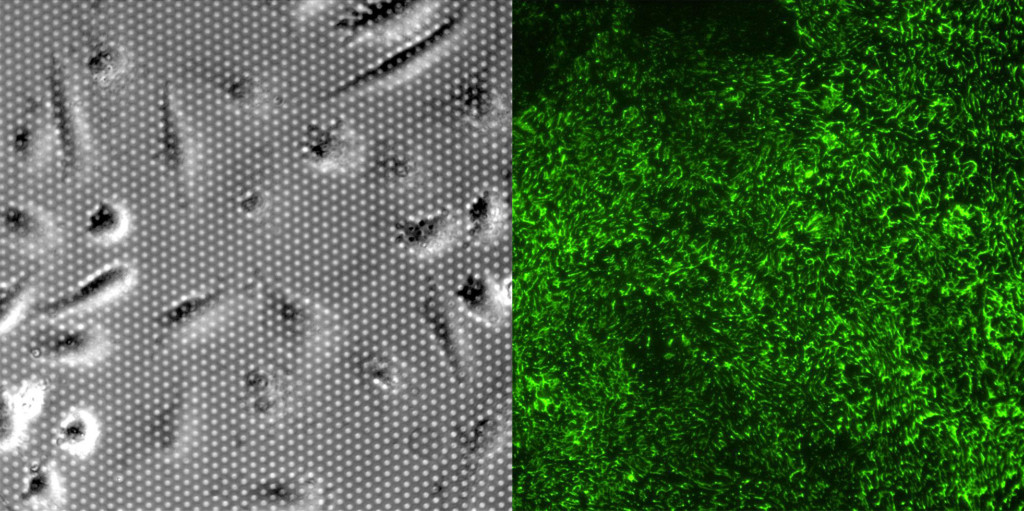
Fibronectin staining was surprisingly robust on both membrane and TCP substrates at 24 hours. It was clear that the cells left behind “trails” of fibronectin as they migrated on the membrane and TCP substrates. There were striking differences between the substrates. 1) Cells migrated more on the TCP substrate in the first 24 hours, resulting in more fibronectin staining. 2) Fibronectin appears as small fibers. The fiber length is substantially longer on TCP substrates compared to the the porous membrane. It is difficult to discern if the fibronectin is laying over the pores, but the shorter fibers are a likely related to the lower number of FAs on the microporous membranes.
Note: the lack of fibronectin and collage IV staining in cell-free regions of the substrates confirms that the adsorption of a 1% geltrex solution does not result in significant deposition of these proteins relative to the ECM that is ultimately deposited by the cells.
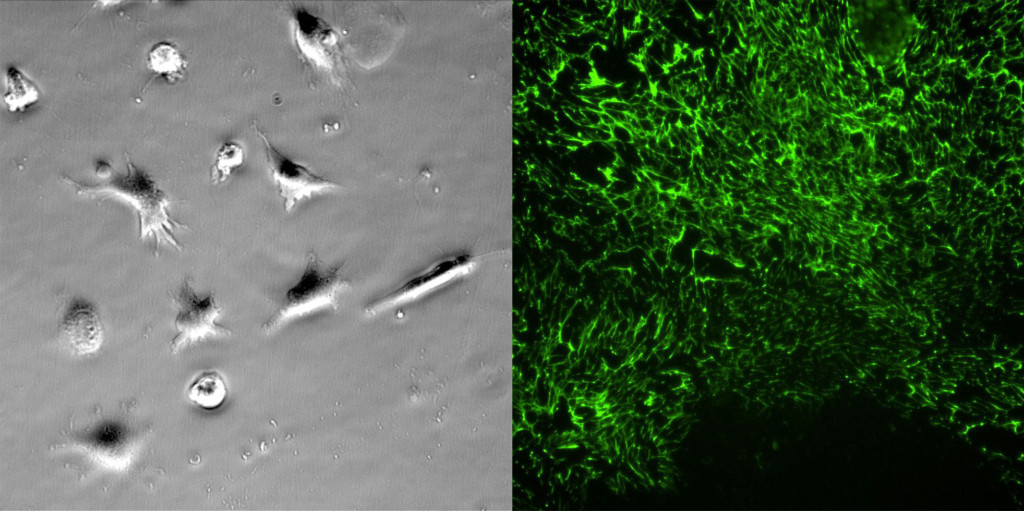
On Monday we will be staining cells at 6 days. We expect more robust fibronectin staining and possibly increased collage IV. We’ll update the post after collecting those images.
[UPDATE July 6 2016]
We didn’t see much difference in Fibronectin assembly at 6 days compared to 1 day. In review of the literature, it seems that most people report FN at 24 hours. Collagen IV was not as well stained and it seems prudent to stick with one ECM protein – FN seems to be a favorite. We have found that soft and stiff PDMS also have short fibronectin fibrils and this matches results in the literature from several groups that have concluded soft substrates result in less FA fibrillogenesis. We were also able to capture an image of a 3.0 um pore size membrane at the non-porous border. It’s clear that longer fibrils are present on the non-porous region and extend slightly into the porous region.
Here is the figure we plan to use in the manuscript:
