Exosome Scale Experiments
Hello Everyone,
This is another quick update over our experiments.
Exosome Scale Experiments:
In all of our experiments, beads are 100nm and pores are 80nm. As you probably remember, in my last post, i talked about hos the membranes are resisstive and they don’t stand these flow rates, so the flow finds another source in the system to leak. So we decided to try different ways to glue the system or bond the membrane and the silicon layers.
Experiment 1:
In this experiment, we used pre-cured PDMS to bond the membrane and the silicon layers, and the result was not bad. we were able to successfully capture beads, and assuming that the features that we are seeing are beads, for the first time in my exosome scale experiments over TE membranes, we saw beads getting paused on the membrane as you can see in this video. The experiment condition was 10 microliter/min pushing and 2 microliter/min pulling.
The next video is the edge of the membrane, so basically, the region which is applied to the transmembrane pressure, is capturing beads but the region next to it without any transmembrane pressure is dark and is not able to capture beads. So we are showing by this video, that only the area with transmembrane pressure on it, is able to capture beads.
Unfortunately, the releasing was not successfully done.
Experiment 2:
So we found a bonding protocol in a paper, and i searched for citations and finally found the first paper using this protocol (papers tittle: Leakage free bonding of porous membranes into layered microfluidic array systems). The process is shown:
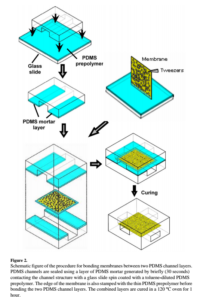
We fabricated some devices and tested them, if the mixture leaks to the channel for any reason, after curing, it blocks the channel and eventually causes leaking in the system. one of the devices survived and i could run it successfully. The experiment condition was 5 microliter/min pushing and 1 microliter/min pulling.
Capturing Step:
It is important to mention that, in these experiments, when i was pushing, we were getting the flow from the top channel also. I was pushing by 5 microliter/min and pulling by 1 microliter/min, but when i started the pulling, the membrane wasn’t able to let 1 microliter/min flow to the top channel from the bottom channel which is the reason for getting some flow from the clamped side of the top channel. But here are my arguments and thoughts over releasing and capturing using pulling and pushing:
- With just pushing: I saw beads that got stuck: even without pulling, transmembrane pressure is the reason for beads that get stopped in the flow by the membrane.
- With pushing and pulling: Transmembrane pressure is probably larger compared to just pushing system (my next experiment), and the fact that we saw the flow was coming from the top channel also, and being able to see beads getting stuck by the membrane show that the membrane is not that permeable. (The membrane is not permeable enough for this flow rate)
Capturing Step:
Intensity out of interaction area: First order of non-specifically absorbed beads
Intensity from interaction area: Captured Beads + First order of non-specifically absorbed beads + Second order of non-specifically absorbed beads.
Furthermore, we need to define capturing for ourselves, so we basically can have 2 types of capturing, first having a bead perfectly and completely sitting on a pore, and also a bead that is partially sitting on the pore and partially non-specifically bonded.

Releasing Step:
Signal before releasing – signal after releasing = Captured beads,
Therefore ideally those beads that are still on the membrane and not released, are non specifically captured.
So the fact that we couldn’t release the beads can be due to two reasons:
- Those signals are not from captured beads and they were from non-specifically absorbed beads
- The membrane is not permeable enough and we are not able to push the flow through the membranes or not strong enough which is needed to release the beads
So my hypothesis is capturing doesn’t mean perfectly having a bead sitting on the pore, it can be partially on the pore. So basically, for capturing, beads have to feel the transmembrane pressure and get captured, and if this capturing is partially, that means for releasing them, we need to have strong enough flow through the membrane, and not just by reversing the transmembrane pressure.
In addition to all of these, we have a good control right now on figuring out the leaking. Anytime we are not capturing or releasing, we definitely have a leaking problem in the system, which means the transmembrane pressure is not applied on the membrane. We can also find the leaking site using nanoparticles.