Update: Critical Flux Experiments, Trial and Error
Hi everyone, in this update I’m going to go over some of the highlights from my experiments over the last month.
After going through several trials that ultimately ended in failure, we moved over to testing long term performance of a 2.7% porosity nano-porous membrane (avg. pore diameter of 33.2 nm) by assessing it in 45 minute steps for controlled flow conditions. Specifically, every 45 minutes (2700 seconds) the ultra-filtration rate increased by 10 percent of the supply rate. So if the supply rate is 30 ul/min, then the first ultra-filtration rate is 3 ul/min for the first 45 minutes, 6 ul/min for the second, and so on and so forth. First I’d like to present controls for 30 ul/min supply rate flow.
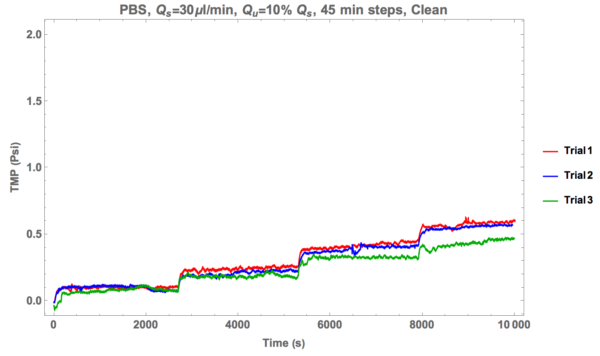
Now onto the data.
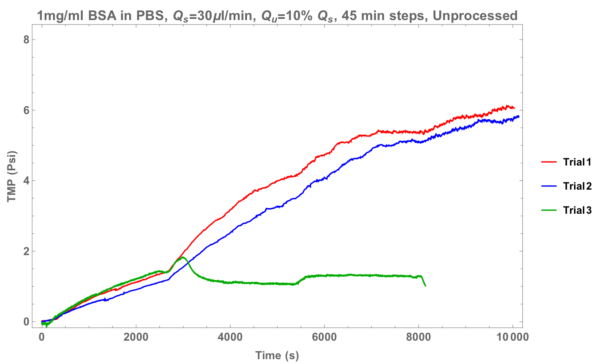
Above is a graph that details some of the first data sets I acquired using this new criteria. In an interesting development, trial 3 (green line) had stable trans-membrane pressure for varying steps. However, all other trials behaved similarly to the red and blue lines as seen above; the membrane kept fouling and achieving non-fouling performance rarely occurred.
As time goes on in these experiments, a protein cake layer builds on the top channel side of the membrane. We speculated that perhaps we managed to successfully shear this protein cake layer off the top of the membrane in the green line trial, resulting in open pores that would allow protein through to the bottom channel. To test this, the supply rate was increased to 60 ul/min (with the resulting ultra-filtration rates being 6 ul/min, 12 ul/min, 18 ul/min, etc.). Here are the results:
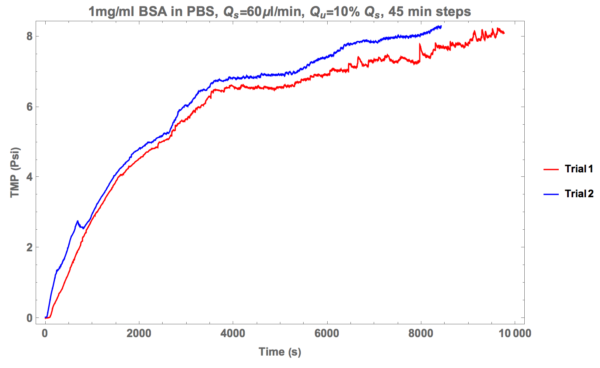
These are mediocre results. After two trials it became abundantly clear that this was not going to result in an acceptable solution.
We abandoned the idea of attempting to shear the cake layer and instead decided to test and see if pulling harder (via ultra-filtration) would result in more pores clearing. To do this, tests were conducted with 1 mg/ml BSA in PBS with a supply rate of 30 ul/min. Ultra-filtration was set at 10% initially and then increased by 20% every step.
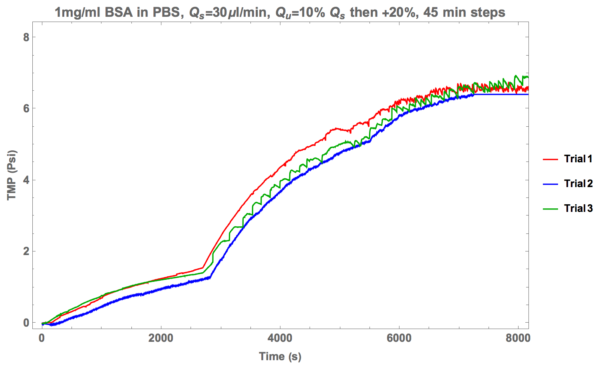
Not quite what we were hoping for. Unfortunately the fouling is worse in this case.
Having assessed some mechanical options, the next choice was to work on the fluid itself. Dr. McGrath and I devised a plan where we would use three different methods and assess how well they performed.
-Filtering
-Open Reservoir
-Ultracentrifugation
First lets look at what happened when I ran my BSA in PBS through a 0.2um filter.
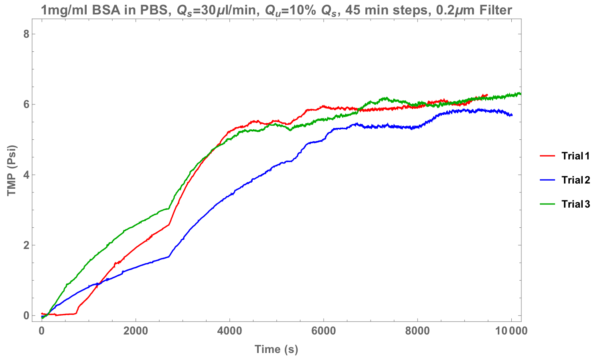
Again, we see fouling. Curiously the performance is a bit varied, this might be due to residue from the filter becoming embedded in my solution.
Now the open reservoir. For this experiment the bottom inlet was un-clamped (it’s normally clamped) and submerged in a 40ml beaker filled with PBS.
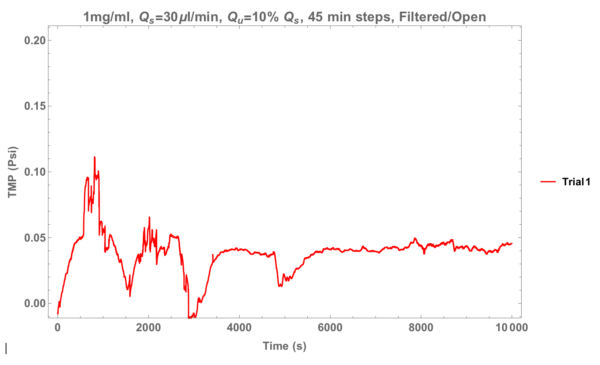
Almost no pressure change. For this set of experimental conditions I expect roughly 1.35ml of fluid to be ultra-filtrated and I noticed that the beaker was missing about this much. Safe to say, this tactic won’t get us further.
Now, take a look at data from ultra-centrifuged (TLS 55 Rotor, 50k RPM, 1hr at 4 degrees C) BSA suspended in PBS (1 mg/ml). Note that a Tecan NanoQuant was utilized in order to assess the concentration of my BSA in PBS, and the result returned was roughly 1.0059 mg/ml.
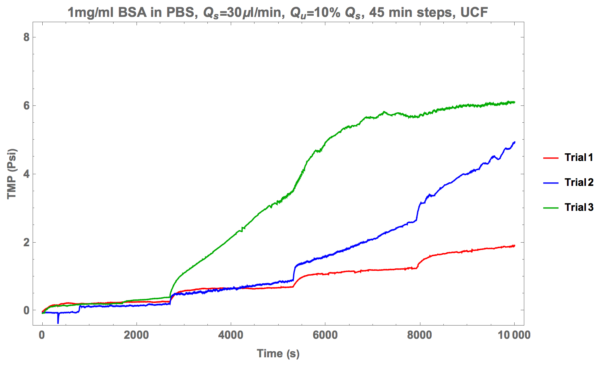
You can see that the first two trials exhibited relatively robust non-fouling performance over a long period of time. Curiously, the third trial (green line) was performed with ultra-centrifuged fluid roughly 24 hours after removal from the centrifuge. We suspect that aggregates formed in my solution and that explains the results I’m currently seeing. Of course, this will be subject to a test in the near future to see if there’s any merit to that claim.
Here’s the same data with controls.
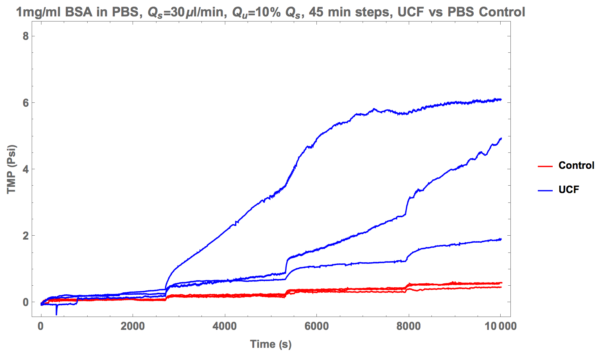
And now here’s the UCF data vs the unprocessed fluid.
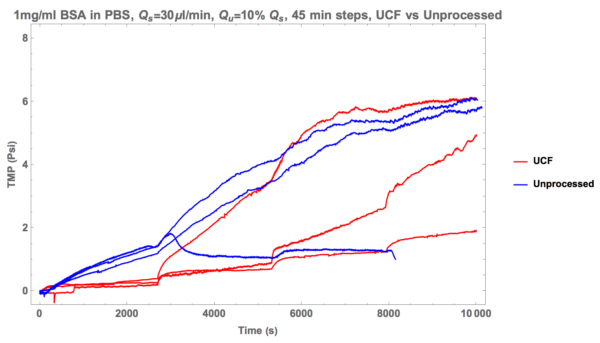 Thus, after many trials, it appears that ultra-centrifugation is the way to go! In the future I’m hoping to share more data with fluid processed this way. The results ought be very interesting.
Thus, after many trials, it appears that ultra-centrifugation is the way to go! In the future I’m hoping to share more data with fluid processed this way. The results ought be very interesting.