BMES 2019 – Cell matrix interactions and platforms for creating engineered ECMs
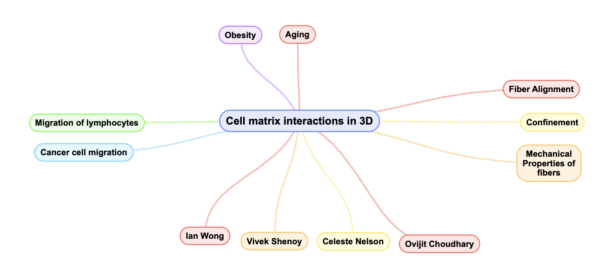
BMES 2019 saw a number of talks being presented on the topic of controlling extracellular matrix architecture and understanding how cells respond to physical cues from their in ECM. My focus was primarily on talks pertaining to the tumor microenvironment or other tissues which show anisotropy in ECM fiber alignment such as cardiac tissue. Recently, there has been a shift from trying to understand the effects of fiber alignment in 3D as compared to usual 2D techniques as is common with micro contact printing, lithography, stamping or other forms of patterning. Figure 1 below shows the major areas of focus along with the key researchers in the field.
1. Programming Cellular Mechanophenotype using Magnetically Aligned Nanofiber Architectures
Alex Hruska, Michelle Adler, Ian Wong
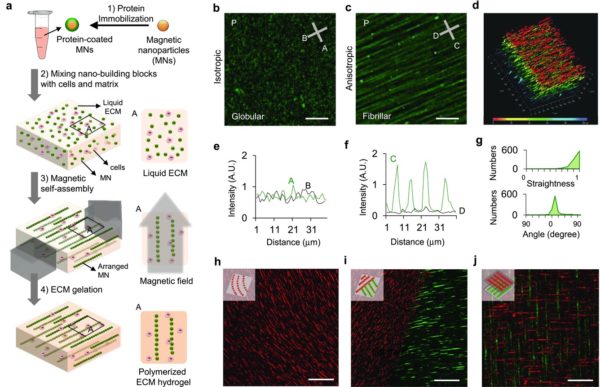
This study aimed to control cell protrusions, morphology and polarization by using ECM fucntionalized, and magnetically assembled superparamagnetic nano beads as a controllable model for understanding the effect of fiber anisotropy on fibroblasts and cancer cells. The fabrication method is shown in Figure 2. They showed that protrusion lengths increased with alignment, and increased concentrations of nanoparticles enabled the formation of more aligned protrusions. Higher alignment also resulted in more directional migration.
Some of the questions this platform raises is:
1. How closely does an aggregate of nanoparticles mimic the actual ECM fiber such as collagen or fibronectin?
2. If we are making a case to understand only fiber alignment, how do we decouple the effects of the GelMA surrounding the cells from the observed effects?
3. To what capacity can cells remodel this ECM?
2. Collagen Fiber Structure Guides 2D Motility of Cytotoxic T lymphocytes
Hawley C. Pruitt, Daniel Lewis, Mark Ciccaglione, Sydney Connor, Quinton Smith, John Hickey, Jonathan Schneck, Sharon Gerecht
In this platform, the authors were aiming to answer questions about T-lymphocyte migration through aligned collagen, specifically in prostate cancer. They were trying to understand whether collagen alignment in prostate cancer guides T-cells to the tumor mass or does collagen alignment inhibit T-cell migration into the tumor. To this end, they fabricated a microfluidic system as shown in Figure 3, consisting of a long 250µm wide channel and a 2mm wide channel, to enable collagen alignment under shear and observed T-cell migration for 20 minutes.

They were able to show that T-cells do not migrate by remodeling the collagen but instead deform between the collagen fibers and move preferentially along the fibers. They observed directional migration and higher velocity in regions with aligned collagen as compared to unaligned collagen. Interestingly, they also mentioned that tumor associated collagen signatures (TACS) as described by P.P. Provenzano and P. Keely (BMC Medicine 2006) might be relevant to prostrate cancer too.
Questions raised by this platform included the following:
1. Do the migration characteristics of T-cells change when being co-cultured with cancer cells?
2. How can we improve the platform to provide interface between the tumor region and surrounding stroma?
3. How does the degree of alignment affect cell migration? Can alignment be shown to be a binary value (yes/no) or is it a spectrum?
3. Collagen Mineralization Increases Breast Cancer Stem-like Properties and Resistance to Doxorubicin
Siyoung Choi, Adrian Shimpi, Joseph Druso, Matthew Whitman, Aaron Chiou, Lara Estroff, Claudia Fishbach
Breast cancer bone metastasis leads to poor clinical prognosis and has been attributed to a small number of cancer cells exhibiting stem cell like properties. CSCs are also found to be self-renewing and present higher resistance to chemotherapy. In this study, the authors studied the effect of mineralized bone ECM on the stem cell like properties of cancer cells, followed by their resistance to drugs. Collagen gel was formed by neutralizing collagen and mineralization was carried out using a solution of salts and acids to form hydroxyapatite crystals between the fibers. Figure 4 shows some of the results

This was a new and interesting area of study for me, a number of other talks focused on trying to understand the role of bone microstructure and mineralized collagen on breast cancer metastasis. Some of the questions being asked included the effect of cancer on bone mineralization and effect of mineralization on microstructure.
The above developed platform can be further developed to include alignment of fibers, interfaces between bone ECM and native breast cancer ECM to understand whether the migration of cells from one ECM to another leads to changes in their mechanophenotype.
Questions we can answer:
- How do the migratory characteristics of breast cancer epithelial cells change when moving from regular collagen to mineralized collagen?
4. Aged Tissue Microenvironment Supports Breast Cancer Cell Phenotype
Gokhan Bahcecioglu, Xiaoshan Yue, Siyuan Zhang, Pinar Zorlutuna
This talk was aimed at answering how age affects the ECM which in turn has an effect on tumor progression of breast cancers. The study was carried out by taking mammary glands from mice of different ages, decellularizing them, quantifying their ECM composition and seeding with MDA MB 231 cells for further studies. While the authors noticed changes in the concentration of collagen V, VI, VII and fibronectin, they also observed significant changes in the ECM microarchitecture. ECM from young tissue explants consisted of thick and long fibers while ECM from aged tissue showed a more wavy morphology with thin fibers. MDA MB 231 culture also showed an up regulation of genes which are commonly associated with increased invasion and migration.

Using the platform developed in our lab, we should be able to provide guidelines on recreating the fiber morphology of aged and young ECM in-vitro.
5. Capabilities of our platform
The platform being developed in our lab can be a versatile tool for overcoming a number of limitations of the above platforms. Our platform offers control over collagen fiber alignment using shear rate and fiber thickness and length by varying nucleation time and addition of molecules such as HA.
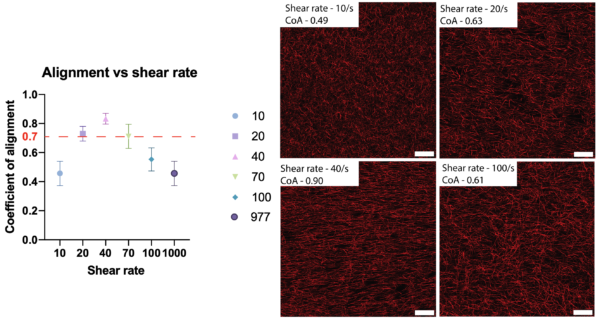
The microfluidic system is based on a modular platform which allows for channel peel-off, offering easy access to the collagen gel within the microchannels with minimal change to microstructure and fiber architecture. This platform can further be supplemented by adding a secondary channel onto the existing collagen network for further analyses such as mineralization, cross linking or creating chemical gradients.

In conclusion, our platform offers opportunities to manipulate 3D ECM gels with the versatility of microfluidic systems and the ease of traditional open systems for studying cell migration across interfaces of tissues.