I’ve looked at cloudiness from both sides now…
“I’ve looked at clouds from both sides now” is a Joni Mitchell song.
Some Background
We are developing a white blood cell (WBC) detector to surveil peritoneal dialysate fluid during fluid exchanges for early detection of peritonitis. This preemptive monitoring is currently not available in clinical practice. The current detection method is by visual inspection of the fluid by the patient who looks for ‘cloudiness’. I’ll briefly discuss the efficacy of the cloudiness-detection method further on but let me remind you that every time you remove a human activity from a process, you remove a chance for error. This alone is enough reason to pursue an automated detection modality.
We are using what is pretty standard for filtering bio-fluids in the McGrath Lab. Specifically, we are using the membranes as a size-selective, fouling-based switch. We are using the presence of We use a three-window 0.5-µm pore membrane chip. Dehghani & Gaborski showed that WBCs could clog a slit filter and using the Ahmed method of clog detection through transmembrane pressure (TMP) monitoring, we will be able to detect WBCs at a level indicating peritonitis (>100 cells/µL). We will look either for the filter to clog in a given time span, or calculate the slope of the TMP increase – if any.
Figure 1: Scanning electron micrograph of white blood cells captured in the pores of a microslit nanomembrane from a 1:1 RBC to WBC mixture. a: 167 X magnification. This demonstrates how WBCs can occlude the microslit pores and cause an increase in transmembrane pressure. b: 473 X magnification. The microslit pores are clearly visible. (Dehghani & Gaborski, Adv Mater Technol 2019)
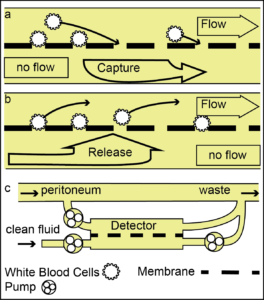
Figure 2: a: Schematic showing WBC-detection switch. Pressure transducers report the transmembrane pressure increase as cells clog the membrane. b: An optional release cycle has been designed to clear the WBCs from the surface of the membrane allowing for multiple measurements during each fluid exchange (drain) c: White blood cells are drawn into the micropores via a transmembrane flow. Transmembrane pressure is monitored to detect clogging.
Device Details:
Figures 3 & 4: Pictures of Julie’s setup.
Top Inlet pushing 200 µL/min
Top outlet is open to the atmosphere (vertical syringe section)
Bottom Inlet is closed (during measurements only)
Bottom Outlet channel pulls 100 µL/min
PDMS Top – 4 punched holes (18 gauge bore- cut off needle)
Top channel Width 3.8 mm Length 10.6 mm
Sealing layer 3.8 x 3.8 mm (around the active area)
3-window 0.5-µm pore membrane (if new membrane I run Corona wand over membrane both sides to make hydrophilic-not necessary but helpful)
Sealing layer 3.8 mm x 3.8 mm (around the active area)
Bottom channel: Width 3.8 mm, Length 17 mm
Glass ~ 12 mm x 24 mm
All channel and sealing gaskets depth are 300 µm
Julie created a guide with all the detailed information about how she runs the experiments.
In Brief, Spent Dialysate from peritoneal dialysis fluid exchanges is pushed at 200 µL/min into the top channel, trench side, with the top outlet open to the atmosphere.
The inlet to the bottom channel is closed and the outlet pulls at 100 µL/min. This, of course, means all the fluid is pulled through the membrane at 100 µL/min (unless the membrane is clogged and the pump can’t keep up.)
We measure and record (thanks LabSmith) the transmembrane pressure for the duration of the experiment at ~0.5-second intervals. We spike the dialysate with varying amounts of white blood cells. We only need to differentiate between dialysate with < 100 cells/µL and >100 cells/µL. (100,000 cells/mL)
Because we are comparing our results to patients detecting cloudiness in their spent dialysate, we don’t need to be 100% accurate. The onset of cloudiness is not always at precisely 100 cells/µL. Additionally, the 100 cells/µL is an arbitrary figure. But those are issues for another research study.
Here is where I get into trouble
We are looking for white blood cells (WBCs). Neutrophils are the most common type of WBC. Neutrophils range from 7.29 µm to 9.68 µm [REF]. We are using 0.5-µm pores so the Neutrophils should at least pause before passing through. Remember we are not using blood. I know you were thinking, what about the red blood cells? NO RED BLOOD CELLS. Hopefully. That would be another issue altogether. But if any red cells block the pores, then signaling the patient to call a medical care professional is a Good Thing (TM).
Preliminary Results
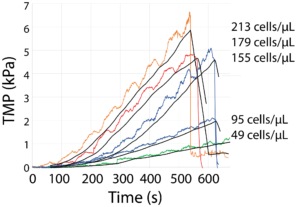
Still much to do… After I’ve gotten a grant (e.g. STTR or R21 if I can fit an RFA, I’ve been advised not to submit another R01 too early)
I need to filter and massage the data to see if there are reliable upturns to detect, or if I can go by the slope alone, which looks pretty good. I obviously need much more data and I need to massage the whole setup to be usable in a clinical setting.
Aim 1: Develop a clinically robust, membrane-based, WBC detector to identify the onset of peritonitis.
Aim 2: Design a point-of-care WBC detector.
Aim 3: Design and carry out a clinical pilot study.