Troubleshooting ECM Protein Coatings on the uSiM
Introduction
The membrane on uSiM platform is inert, therefore, coating with extracellular matrix (ECM) proteins is essential to promote attachment and growth of adhesion cell lines on the uSiM. Besides promoting proper attachment on the membrane, the ECM proteins dramatically impact cell growth, differentiation, and functions. Therefore, careful consideration must be given in selecting the type and concentration of ECM proteins for coatings. In this post, I will be sharing some evaluation and troubleshooting steps when selecting ECM coatings.
My project is to develop an in vitro air-blood barrier model on the uSiM platform by coculturing two primary cell lines on two sides of the membrane: alveolar epithelial type I cells (ATI) and microvascular endothelial cells (MVEC) (Figure 1). I have chosen three candidates for coatings nature to the basement membrane of the air-blood barrier: adhesive protein fibronectin (FN) and fibrous proteins collagen I and IV (Col I and Col IV). I optimized the coating concentration using either one protein or a combination of proteins by evaluating cellular adhesion with the Crystal Violet assay. For my air-blood model, I will be coating membranes on both sides with 5ug/cm2 concentration of FN, equivalent to 18.5ug/ml (100ul) in the luminal and 450ug/ml (10ul) in the abluminal channel. After coating of both channels, MVEC will be seeded on the uSiM in an inverted configuration overnight before it is flipped back for seeding of ATI in the luminal channel. My initial experiments indicated successful attachment and proper morphology and functions for both cell types, however, I recently had some new issues with attachment. There are two factors that were changed in my experiments compared to the initial ones: a new batch of cells from a different donor, and a new batch of FN. After a series of troubleshooting experiments, I have summarized some key points to consider when optimizing ECM coatings on the uSiM: homogeneity of primary cells, differences in the microenvironment of the uSiM compared to standardized culture vessels and pH of the coating solutions.
 Figure 1. Co-culture setup on the uSiM platform.
Figure 1. Co-culture setup on the uSiM platform.
What went wrong?
After setting up a big experiment and finding that my MVEC did not attach to the membrane the next day, I was confused about what could have gone wrong. After careful consideration, I decided to run my first troubleshooting experiment to see if the new batch of cells or the new FN could be the problem. I seeded both of my cell types on both luminal and abluminal channels to narrow down the problematic factor. My results indicated that both ATI and MVEC attach on the luminal channel, but get lifted off and die in the abluminal channel (Figure 2).

Figure 2. Both ATI (A) and MVEC (B) showed attachment in the luminal channel. MVEC (C) showed poor attachment in the abluminal channel. Phase images at 20x magnification, scale bar at 50um.
The results helped me narrow down that the problem is due to seeding in the abluminal channel, which has a much smaller volume-to-area ratio, requiring a higher coating concentration. It could be that this new batch of cells is more sensitive to this (They were expanded in 100mm culture plate with the same FN at concentration of 20ug/ml). The FN comes in stock solution with alkaline pH of 11.0. While the manufacturer indicated that this does not affect cell health when diluted and used to coat tissue culture plates, the differences in culture media volume between culture plate and uSiM are quite significant. Therefore, for my next set of troubleshooting experiments, I explored potential resolutions to my problems: adjusting the pH of coating solution, coating at a lower concentration (20ug/ml, similar to luminal channel), increasing washing post-coating (1x20ul PBS to 2x100ul PBS), add a post-seeding wash step at 2hr timepoint after cells have established initial attachment. The results indicated that lowering coating concentration does alleviate the problem partially, however, also affects the attachment of cells. Washing the cells with media at 2 hour after seeding allowed the channel’s pH to be lowered and attenuated the pH effects on cell health (Figure 3).

Figure 3. MVEC seeded in the abluminal channel of the uSiM. Lowering concentration from 450ug/ml to 20ug/ml affected cell attachment. Washing at 2hr post-seed diminished the effect of alkaline pH on cell health. Phase images at 20x magnification, scale bar at 50um.
While excessive washing solved the pH problems to cell health, time constraints with the 2hr post-wash step could be problematic for a larger sample size. Therefore, adjusting pH of the FN coating solution may be another solution. My results indicated that while this maintained cell health and attachment, this changed the properties of the ECM protein and promote sprouting in endothelial cells (Figure 4). As mentioned earlier, it is important to consider the cells’ BM in vivo and how changes in the ECM can provide different cellular signals to influence cell behaviors.
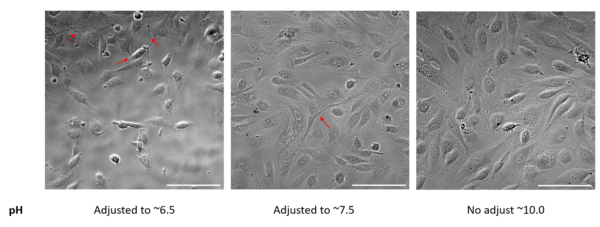
Figure 4. MVEC seeded in the abluminal channel of the uSiM. The pH of coating solution was adjusted using HCl (from ~10-11). Adjustments of pH cause endothelial sprouting (red arrows). Phase images at 20x magnification, scale bar at 50um.
Conclusion
While I have found a solution to my problem, I plan on doing some more experiments to understand why this problem is affecting different batches of cells and different cell types differently. It is important to understand the ECM environment relevant to your cell types, especially primary cells, and take careful consideration in selecting appropriate ECM coatings for the uSiM platform.