Fc Receptor Binding Inhibitor Aggregation Issues in the CAD-LB Platform
Introduction
We are interested in exploring whether certain types of extracellular vesicles (EVs) may be more likely to become trapped while crossing the blood-brain barrier (BBB). Specifically, we are curious if EVs possessing certain types of surface integrins might bind to the vascular basement membrane that surrounds the exterior of brain blood vessels to become trapped. Integrin β1, for example, is known to bind to both laminin and type IV collagen which are the two major structural components of the basement membrane [1]. EVs can inherit the integrins of their parent cells, and it is therefore possible that EVs derived from the hCMEC/D3 human cerebral microvascular endothelial cell line might feature integrin β1 or one of the other characteristic hCMEC/D3 integrins that are known to interact with the basement membrane [2-5]. Our current goal is to use the CAD-LB approach to validate that integrin β1 is present on the surface of hCMEC/D3-derived EVs. Afterward, we intend to perform EV diffusion experiments through pericyte monolayers seeded in the μSiM to see how integrin blockade might affect EV transport (fig. 1).
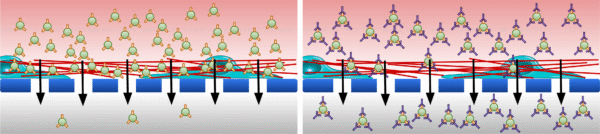
Methods
hCMEC/D3s were expanded in T175 flasks. Upon reaching 100% confluence, the cells were incubated in serum-free media for 24-48 h. The conditioned media was sampled and frozen at -80 °C for long-term storage. Upon thawing, the media was centrifuged at 300xg for 10 min to pellet cell debris and then ultracentrifuged at 100,000xg for 2 h to pellet and isolate EVs. EV concentrations were determined by NTA. For CAD-LB loading, devices with 2-slot NPSN membranes were assembled “trench-up.” EVs were diluted in PBS to approximately 1×107 particles/ml, labeled with 5 μM CFSE (BD Biosciences, Cat. # 565082), and incubated for 15 minutes in a 37 ºC water bath. We subsequently added Fc Receptor Binding Inhibitor (ThermoFisher, Cat. # 14-9161-73) and incubated at 4 ºC for 15 minutes. Samples were then stained with either 200 ng/ml CD29 (Integrin β1) Monoclonal Antibody (TS2/16) conjugated to APC (ThermoFisher Scientific, Cat. # 17-0299-42) or 200 ng/ml Mouse IgG1 kappa Isotype Control (P3.6.2.8.1) conjugated to APC (ThermoFisher Scientific, Cat. # 17-4714-42) and incubated with gentle mixing for 45 min at room temperature. The lower channel of the CAD-LB devices was blocked with 1 mg/ml BSA for 15 min. PBS was added to the top well and one of the lower channel access ports was covered with double sided tape (3MTM, Cat. # 468MPF) to create a “dead-end-like” filtration flow path. 40 μl of sample was injected through the open access port at a controlled rate.
Results and Discussion
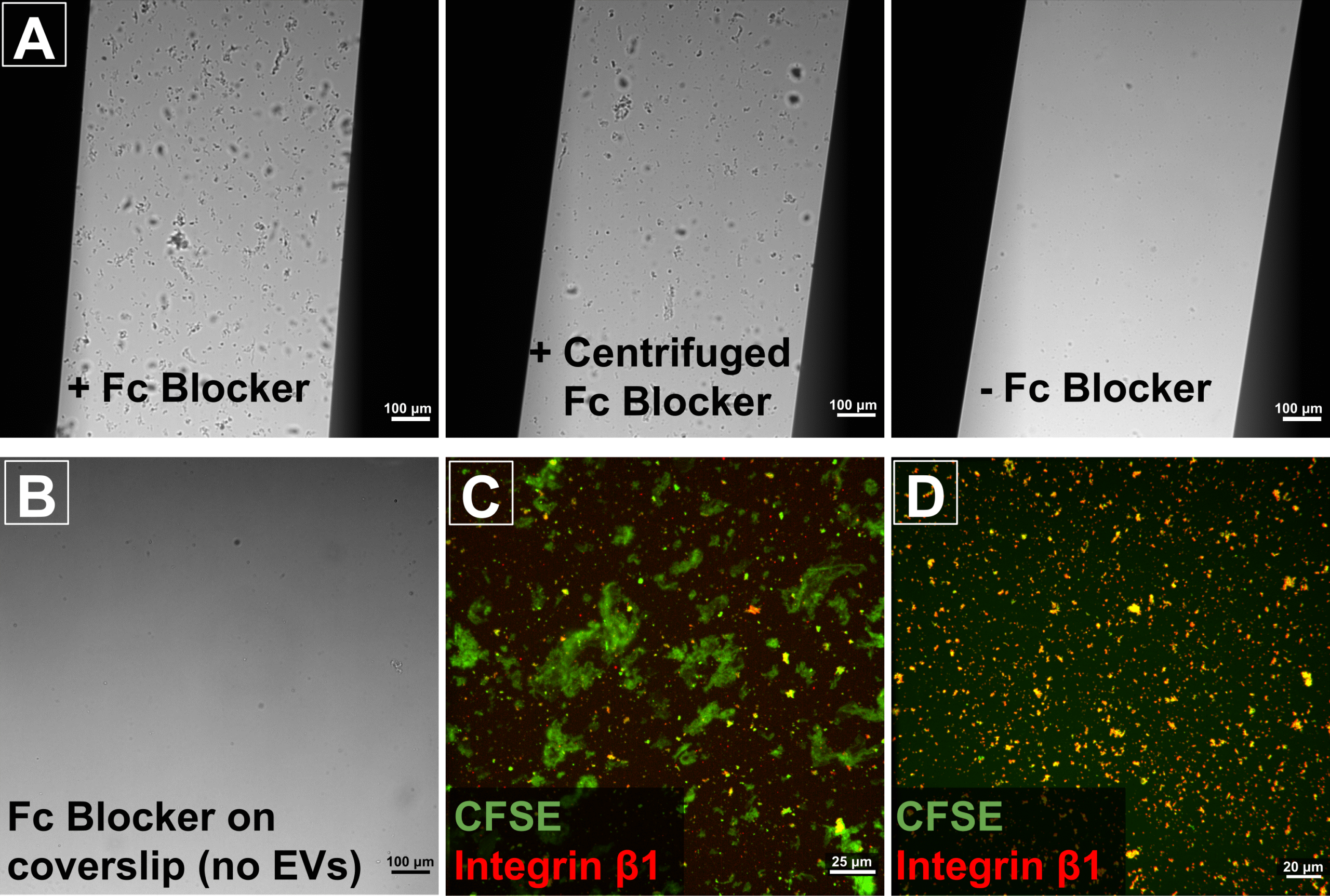
While we had some initial promising results, we were hampered by the appearance of many large aggregates on our membranes. We tested every component of our preparation. This included the age of the assembled CAD-LB devices since we had been warned that the pressure sensitive adhesive could potentially spread across the chip given sufficient time. However, the culprit turned out to be the Fc Receptor Binding Inhibitor. The purpose of including this inhibitor was to reduce nonspecific binding of our antibodies and isotype controls, so we hoped that we would still be able to incorporate it into our experiments. However, centrifuging the Fc Receptor Binding Inhibitor at 16,000xg for 20 min failed to completely remove the aggregates (fig. 2A). Further investigation demonstrated that the Fc Receptor Binding Inhibitor solution itself did not contain noticeable aggregates (fig. 2B). Rather, the aggregates seemed to form due to an interaction between the Fc Receptor Binding Inhibitor and the EVs in our samples. This was substantiated by the fact that the aggregates appeared to contain the CFSE fluorescent label that was intended to serve as a pan-EV label in our experiments (fig. 2C). Filtration after labeling could theoretically remove these aggregates to produce cleaner CAD-LB images, but there is a potential risk that doing so might remove a subset of EVs from the analysis. Going forward, we have opted to omit the Fc Receptor Binding Inhibitor from our experiments. This may increase the risk of non-specific binding of our antibodies, but seems worth it in order to preserve the whole EV population. We have begun performing CAD-LB experiments without the Fc Receptor Binding Inhibitor, and the quantity and size of apparent aggregates has been greatly reduced (fig. 2D).

We intend to repeat the integrin β1 labeling experiment again to complete the dataset so that we can determine if colocalization with the CFSE-stained EVs is higher with the anti-integrin β1 antibody compared to the isotype control. This will verify that the hCMEC/D3s feature integrin β1 on their surfaces. We are also interested in following this up by examining expression of α3 and α6 integrins which can also interact with the basement membrane. If we are convinced that the hCMEC/D3-derived EVs express one or more of these surface adhesion molecules, we will proceed with the aforementioned diffusion studies to examine particle entrapment at the BBB.
References
[1] T. Osada, et al., “Interendothelial claudin-5 expression depends on cerebral endothelial cell-matrix adhesion by β(1)-integrins,” Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism, vol. 31, pp. 1972-1985, 2011.
[2] V. Dozio and J.-C. Sanchez, “Characterisation of extracellular vesicle-subsets derived from brain endothelial cells and analysis of their protein cargo modulation after TNF exposure,” Journal of extracellular vesicles, vol. 6, pp. 1302705-1302705, 2017.
[3] Y. Feng, et al. (2022, The Blocking of Integrin-Mediated Interactions with Maternal Endothelial Cells Reversed the Endothelial Cell Dysfunction Induced by EVs, Derived from Preeclamptic Placentae. International Journal of Molecular Sciences 23(21).
[4] M. A. Lopez-Ramirez, D. K. Male, C. Wang, B. Sharrack, D. Wu, and I. A. Romero, “Cytokine-induced changes in the gene expression profile of a human cerebral microvascular endothelial cell-line, hCMEC/D3,” Fluids and Barriers of the CNS, vol. 10, p. 27, 2013/09/19 2013.
[5] A. N. Stratman, K. M. Malotte, R. D. Mahan, M. J. Davis, and G. E. Davis, “Pericyte recruitment during vasculogenic tube assembly stimulates endothelial basement membrane matrix formation,” Blood, vol. 114, pp. 5091-5101, 2009.
Here is a link to a Thermo fluorescent dye labeling kit that includes the equation for calculating labeling ratio: https://www.thermofisher.com/document-connect/document-connect.html?url=https://assets.thermofisher.com/TFS-Assets%2FLSG%2Fmanuals%2FMAN0019835_AlexaFluor_ProteinLabelingKits_UG.pdf