Organic-Inorganic Hybrid Membranes Formed from Block Copolymers and Titanium Dioxide Nanoparticles (A Review)
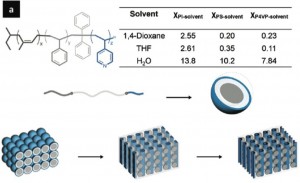
The Wiesner lab at Cornell (whom you may remember as the discoverer of the neutrally charged but highly fluorescent “C-dots” Jess used for her charged-based separation work) are about to publish Asymmetric Organic-Inorganic Hybrid Membrane Formation via Block Copolymer-Nanoparticle Co-Assembly in Nano letters. Building upon their previous work on block copolymer self-assembling films, they have added titanium dioxide nanoparticles to the polymer mixture with the hopes that eventually the inorganic nanoparticles will contribute mechanical and thermal stability, chemical activity, and pressure resistivity. They offer no data that the presence of nanoparticles does affect any of these properties, but they do show rather conclusively that the titanium dioxide is actually incorporated into the membrane. Additionally, the permeability and structure of the membrane change with nanoparticles added.
Diblock and triblock co-polymer-based porous membranes form by microphase separation. A typical diblock co-polymer will have a highly soluble polymer covalently bonded to a much less soluble polymer.
If a mixture of the co-polymer and solvent is extruded as a film, as solvent evaporates the co-polymer will form nano-scale micelles which then pack in a cubic or hexagonal lattice. These lattices have vertically aligned pores, the size of which can be tuned by varying the length of the co-polymers.
Early work in these films used thick films for mechanical stability, but these had long pores and low permeabilities. Another group used thin porous films that were then transferred to a support membrane. The Wiesner group’s innovation is to use triblock co-polymers (which add another degree of control) and another regime of film formation within the same film to produce a support layer that does not contribute as much to the resistance of the membrane. Whereas slowly letting solvent evaporate from the top of the film allows highly monodisperse self-assembled nanopores in the range of 16-30 nm, rapidly replacing solvent with water (a process called “nonsolvent-induced phase separation”, i.e. dunking the film in water) causes a “macroporous support structure” to form. The film formation takes three steps:
1. Grade the wet film (triblock copolymer and solvent) out to a standard height (225 in this case)
2. Wait some tens of seconds for solvent to evaporate and nanopores to self-assemble
3. Rinse with water
The results are impressive:
The group reports hydraulic permeabilities of up to 274 , or 0.457
. Typically our pnc-Si permeabilities are ~10
. When titanium dioxide nanoparticles were added to the film, the spongelike macroporous support structure in the back changed form substantially, forming large tunnels:
These large “fingerlike” pores do not impede flow as much as the purely organic spongelike supports, leading to very high permeability: 3200, or 5.3
, which is an order of magnitude better than the purely organic membranes and just half that of pnc-Si. SEM images reveal that the membranes with Titanium oxide suffer from lower pore density, so if the group can fix that problem it seems likely they will have permeabilities equalling or exceeding that of pnc-Si.
I’m not positive I’m reading the stress-strain graphs correctly, but it seems like the membranes have a burst pressure of 3.5 MPa, or ~500 psi:
(for purely organic membranes)
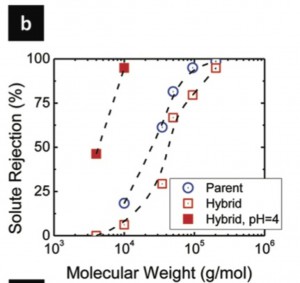
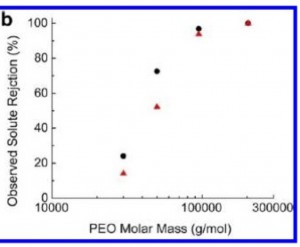
Finally, the membranes have good separation characteristics.
Organic cutoffs:
Hybrid cutoffs:
To summarize, this membrane is stronger than both pnc-Si and nitride, has comparable permeabilities and separation characteristics, is (presumably) cheaper and easier to construct, and it’s formation is reasonably well understood. However, pnc-Si and nitride are four orders of magnitude thinner (copolymer membranes are ~220 m thick), are tuneable over a wider range of pore sizes, and are totally inorganic. We should definitely keep an eye on these guys.
UPDATE: Here is the supplemental information for the paper. After discussion at NRG on 12/10/13 we came to the conclusions that the group is sacrificing the surface uniformity, lack of pinholes, and monodispersity of pore sizes they achieved in the first paper for the sake of higher permeabilities. They are still worth watching, but they won’t be competitive with our nanomembranes without some substantial development.




They claim … “These hybrid membranes exhibited a defect-free
surface over relatively large areas (at least 25 μm2 as shown in
Supporting Information Figure S3) and the pore sizes were
nearly monodisperse.” We should upload their supplement to you post so we can review.