Adsorption to polymer membranes
Previously we have studied protein adsorption to pnc-Si membranes by TEM and fluorescence. Neither of these methods will work with polymer membranes. Polymer membranes are too thick to view using TEM. Polymer membranes also autofluoresce creating a lot of noise while using our fluorescence method. Here I outline a new method of studying adsorption to polymer membranes.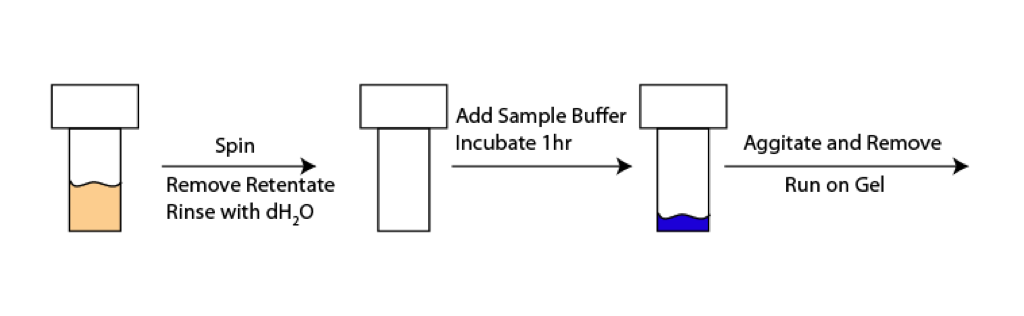
As illustrated in the scheme above, I spun Microcons and Nanoseps with 1mg/mL BSA. These samples were spun at 14000g for 10min, as indicated in the manufacturer’s specs. After the spin I removed all of the retentate, and washed with dH2O. I then added 50uL of 1x Sample Buffer to the device. Sample Buffer is commonly used in protein gels to denature proteins. The idea is that in this case it will denature and lift the adsorbed proteins off the membrane. I allowed the sample to incubate for 1 hour, and then agitated it with my pipette and removed the Sample Buffer. I ran the Sample Buffer on a gel, see below.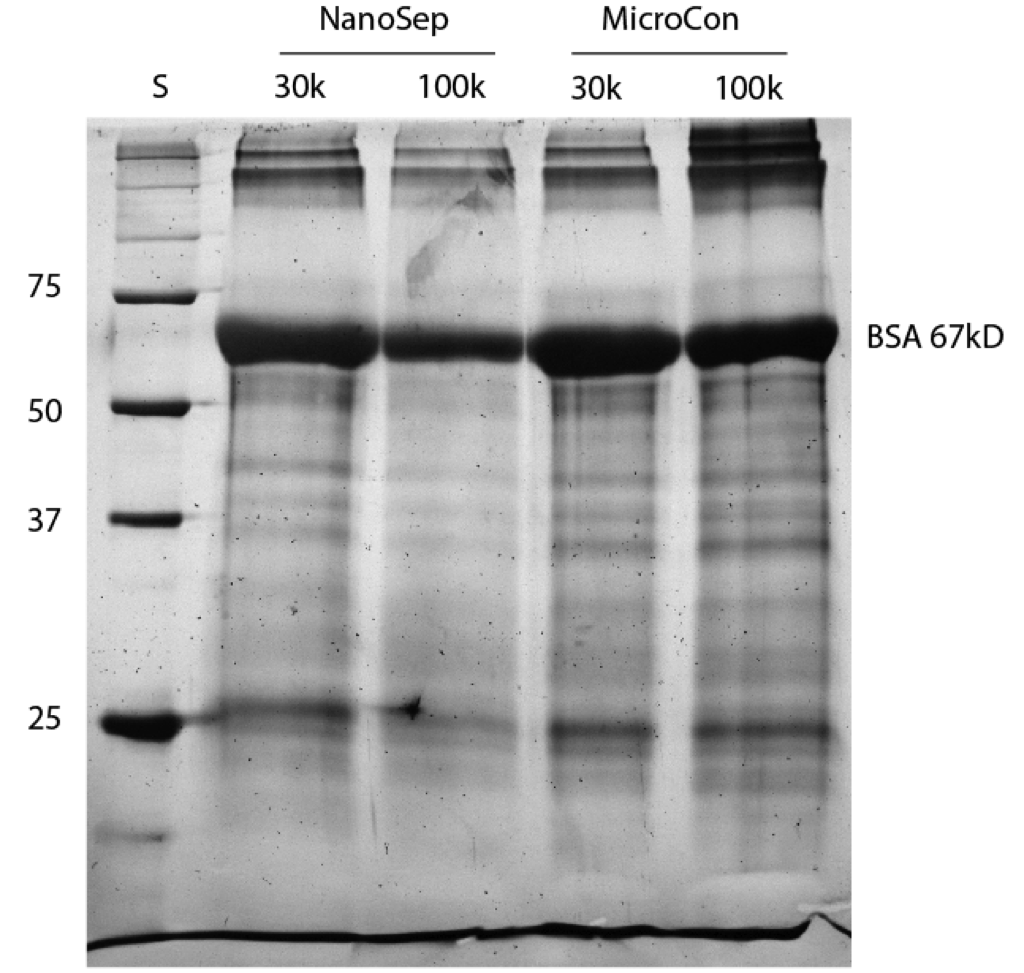
First off, this BSA sample isn’t incredibly pure, so there’s other bands in this gel besides BSA. We do see that even after a dH2O wash that there’s still a lot of protein remaining on the membranes. There’s a little less on the NanoSep 100k rather than the 30k, which might be due to the fact that more BSA is able to pass through. This isn’t necessarily the best way to view adsorption in these types of membranes, but it seems to work. Tom and I were also discussing the possibility of spinning the sample buffer a bit through the membrane in order to pick up some of the protein stuck more deeply within the polymer membrane, which isn’t necessarily an improvement on this method. One could also dissasemble the spin tube and immerse the membrane in sample buffer for a while, but there is the problem of dilution and detection so only a little sample buffer can be used.
The next step would be to see if we can get a similar picture with the pnc-Si membranes. There is the different surface areas to consider, but it might be possible to come up with a similar gel.
It gives us signal and so I think its the best way. Even if the measurement doesn’t capture all the protein lost to the membrane, the comparison to pnc-Si should be conservative and possibly dramatic. We don’t need perfect quantification if the differences are night/day.
I would have guessed that microcon cellulose would bind less protein the nanosep PES based on some discussion I had with Millipore. The difference may be related to better retention of protein behind the Millipore devices at the same size.
We have to wait for new material to compare to pnc-Si by convection, but we could do the passive adsorption relevant to diffusion with any shards of pnc-Si we have laying around the lab.
Good idea.
I was impressed with how easily this worked. Hopefully we can work out a good comparison with the pnc-Si.
Is there any way that this can be even semi-quantitative? If the adsorbed protein is less than 1%, I doubt anyone would care, so the comparison looses significance. If this is 5% or more of the total sample, then there is something to be said.
I think both manufacturers report retentate recovery of 99%, right? If we are seeing substantially more, we should contact tech support for more insight.