Cell Adhesion Study (Trial 2)
I repeated the previous experiment, this time plating slightly higher density of cells: ~15100 cells/sq.cm. Samples were from the same W673, no RTP)
After counting the cells stained using Calcein AM, % Adhesion w.r.t the cells plated were found to be:
non-silanized samples = 55.87%; silanized samples = 41.33%
In above figures, not many cells are found in the active area. This could be due to the low density of cells or breaking of membranes. Also, the good news is that the % adhesion values for this format (3D) are nearly the same as that of 2D format. One more successful trial would show if there is any significant difference between the silanized and non-silanized samples in terms of cell adhesion. (Keeping in mind that proteins from serum could coat the silanized surface making it similar to the non-silanized; this needs to be verified through contact angle measurements.)
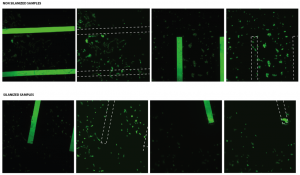

Again, the cell morphology looks weird. It could be because this is only a few hours after plating, and thus the cells are somewhere in between their spherical (non-adhered) and “normal” morphology.
I think that if the membranes are broken when you are doing the imaging, you should throw that sample/data away. If the membrane broke before cells adhered, we don’t know how many (if any) cells got through the hole in the membrane. This could significantly decrease the effective initial cell seeding density, thus changing the % adhesion.
If two samples have their membranes broken completely, doesn’t this mean that the no. of cells adhered to both of these be probably the same? (can’t it be assumed that the no. of cells lost through the slits is probably same in both cases)?
It does look like the cells on the silanized samples are more rounded (some look necrotic). We have image processing tools to extract the area of a cell and quantify the degree of cell spreading. These cells need to spread to grow and so growth data will indirectly reveal problems with silanization, but I would be in favor of quantifying the spreading directly. Contact angles first. In the meantime we might want to get some new HUVEC from the medical center or work with 3T3s for a while.
Anant,
I think you need to account for the loss of area caused by the broken membranes. Without the membranes, the cells can’t attach and those areas in the microscopy images are like a “dead” area. I think % cell adhesion is being underestimated in those images.