Carbonization can occlude pores
Last week I reported that carbonization can shrink pores. This week I was able to completely occlude the pores with a change in the process recipe. Instead of cutting off the acetylene flow before the thermal treatment, I kept the gas on for the entire process. What resulted was a complete occlusion of the pores.
The recipe I used:
- Place membrane in susceptor
- Flush RTP chamber with 10 LPM Ar for 5 min at 20 C
- Flush RTP chamber with 8 LPM Ar and 2 LPM C2H2 for 1 min at 20 C
- Heat chamber to 800 C with 2 LPM C2H2 and 8 LPM Ar for 5 min
- Cool chamber down for 10 min with 10 LPM Ar
I decided to try the process without the pre-treatment HF dip to see if a reaction would still occur. The literature states that there must be Si:H available on the surface in order for the carbonization to occur. I’ve always thought that our membranes did not have a high enough density of hydrogen at the surface so I pre-treated with HF, but it appears the SiC still goes down without the extra HF dip.
Now that we’ve shown that carbonization can completely block pores, it is possible that by tuning a) the RTP temperature b) the treatment duration and c) the concentration of acetylene we can control the degree of occlusion.
As a control, I put a membrane through the same recipe described above but without the acetylene flowing.
There appears to be no change between the no treatment and RTP only sample.
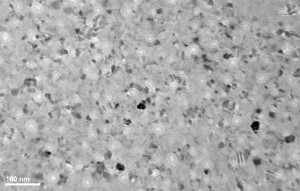
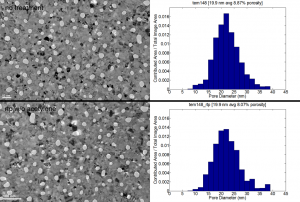
this is interesting and nice. Of course, the real cool stuff would be full control of the cutoff pore size with carbonization. You could think of this as “trimming” the pore size. This result suggests that carbonization leads to a solid film, not just chemical molecules on the sample’s surface. I’d love to have them analzyed for chemical and eventually crystallographic properties….