Gold particle update
Previously, I had data showing that 5nm gold in 10mM salts went diffused through pnc-Si membranes. I was able to achieve this twice, although the second time took two days before the gold signal was intense enough to observe via absorption. Here’s what has happened recently:
- 5nm gold will not pass through untreated SC 168, observed for 2 days, but other membranes in this series were discoloring and breaking
- 5nm gold will not pass through RTP’d SC 168 – one day and counting
- 5nm bsa-au (10nm total size by DLS) does not pass through RTP’d SC168, one day
- Rhodamine readily passes in one day
- This series of wafers seems to have higher charge than previous wafers. This is evidenced by faster EO of unoxidized SC166.
I plan on trying to modify the membrane either by aminosilanization or BSA adsorption and try separations with 5nm au in 10mM KCl. Carbonization may be worth a try too. Also, I have been thinking about driving the separation by the osmotic flow that the Paul mentions in his post.
I am receptive to any other ideas for reducing membrane charge or passivating the gold.
Previous membrane that 5nm gold passed through:
The membrane from these experiments:
The were some elongated pores for the old wafer that led to the larger pore sizes, but they are few in number. The density is skewed to the smaller pores. The new wafers have high densities of 30nm round pores, and my guess would be that the charge characteristics are playing more of an effect in these separations.
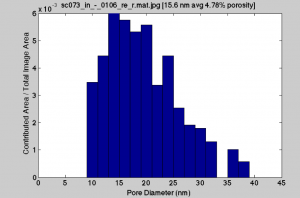
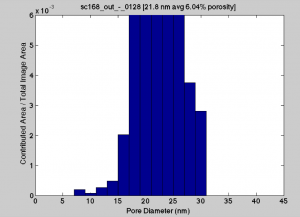
Grrr …
Do we know what the relative pore sizes are for the membranes that do/do not work?
If it’s a membrane charge issue then silanization is the way to go…
I’m pretty intrigued by the carbonization idea. If carbonization can be used to knock down membrane charge, we might be able able to realize the cut-offs suggested by TEM images.
The gold is not aggregating. However, I can’t probe higher salt concentrations than 10mM because it will aggregate.
I have functionalized the gold; the BSA-Au sample is coated with BSA. This raises the size by 5 nm, but allows us to use salt concentrations up to 100mM. The fact that this did not pass is either due to 1) the new size is too big, 2) there is still too much repulsion even in high salts and a lower charged gold colloid (the bsa coat reduces surface potential), or 3) some sort of desorption/adsorption interaction with protein and membrane. When you reduce surface potential of the colloids with a coat, you need a physical barrier to stop the particles from aggregating. The small ligands I’ve used to functionalize gold have all led to aggregation. BSA is the smallest I can get and retain colloids.
The preliminary results (from contact angles) indicate that the carbonized membranes have about the same charge as RTP’ed membranes.
How about functionalizing with a medium-sized ligand – bigger than citrate/other surface groups but smaller than BSA? Maybe a short PEG?