Protein Assay Test
Over the past few weeks, I have been trying to separate proteins with the SepCons. As a preliminary test, Barrett and I decided to first acertain that BSA could pass through a membrane with pinholes. Theoretically, the pinholes are so large that the BSA should easily pass through. However, it doesn’t appear that the BSA is passing through like it should be. This post is a summary of the experiments I have done and their results. If anyone has any suggestions or ideas about why it isn’t working, please let me know!
6/4/2009: BSA Test #1
Wafer SC 052
5 tests:
- PBS straight from the bottle
- pncSi with pinholes, 2mg/ml BSA in apical (positive control)
- unetched pncSi, 2mg/ml BSA in apical (seal test)
- pncSi with pinholes, PBS in apical (negative control)
- PET, 2mg/ml BSA in apical (positive control)
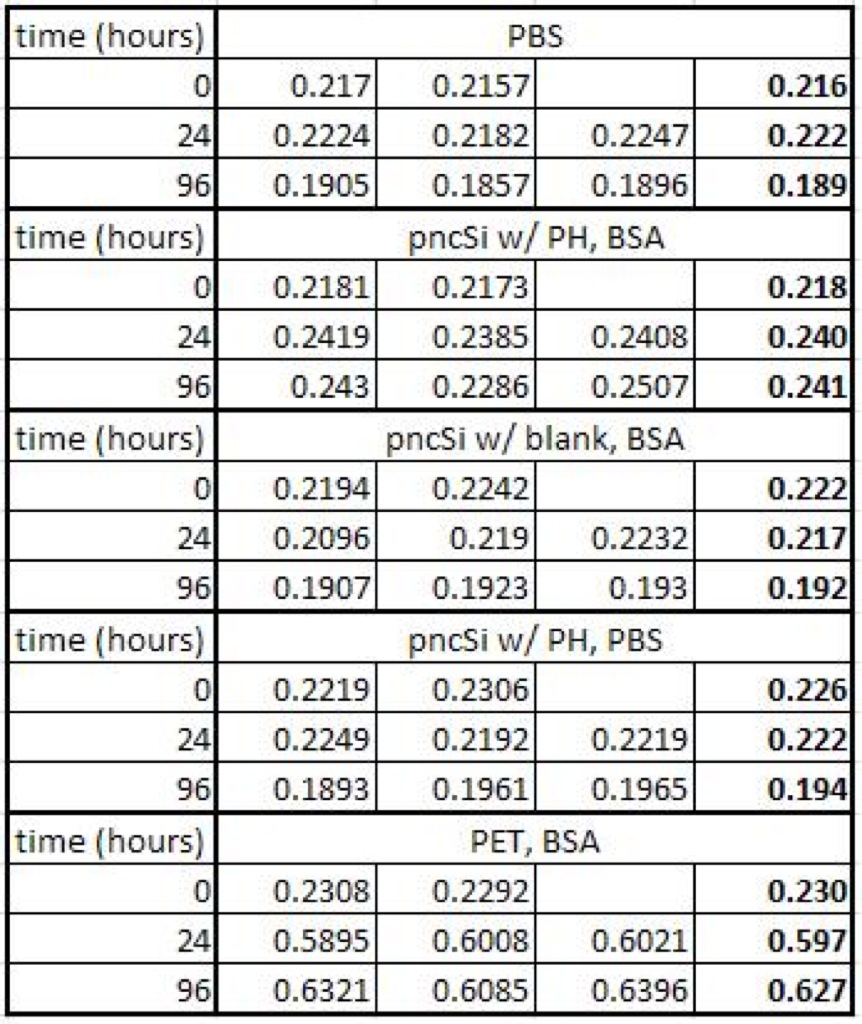
Absorbance Data (the bold numbers are the averages):
The PBS, blank chip, and pnSi with PBS in the SepCon absorbances hover around 0.21 abs. The pncSi with BSA shows a slight increase of only about 0.02 abs between hours 0 and 24 and then levels off.
6/8/2009: Test a purposefully torn membrane to see if something with the chip is preventing the BSA from going through
Wafer SC 052
After 24 hours, I added the diluted dye to a sample and it turned bright blue. So, the protein went through.
6/9/2009: BSA Test #2
Wafer SC 074
2 tests:
- membrane with pinholes, PBS
- membrane with pinholes, 2mg/ml BSA
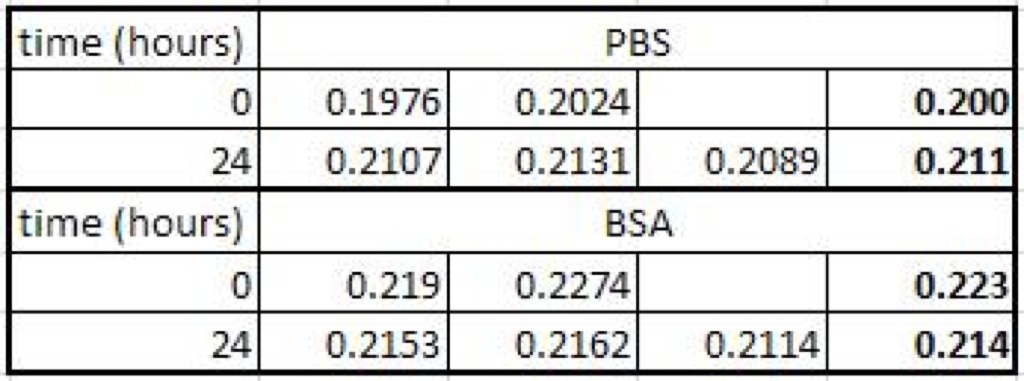
Absorbance Data:
After the reading at 24 hours, I put them on the shaking plate. I tested the filtrate again at 48 hours, but there was no hint of blue, so I didn’t bother measuring the absorbance with the TECAN.
6/11/2009: Avidin Test #1 (is something weird with BSA?)
Wafer SC 099
After 24 hours, I added dye to a sample and there was no trace of blue.
6/23/2009: BSA Test #3- with DI H2O in the basolateral, instead of PBS
Wafer SC 086
After 24 and 48 hours, there was no trace of blue.
6/29/2009: Protein Assay with Fluorescine
Wafer SC 214
3 tests:
- membrane with pinholes, 1:1 mixture of 10mg/ml BSA in HBSS and 0.02% fluorescine in apical
- membrane with pinholes, 0.01% fluorescine in apical
- just HBSS
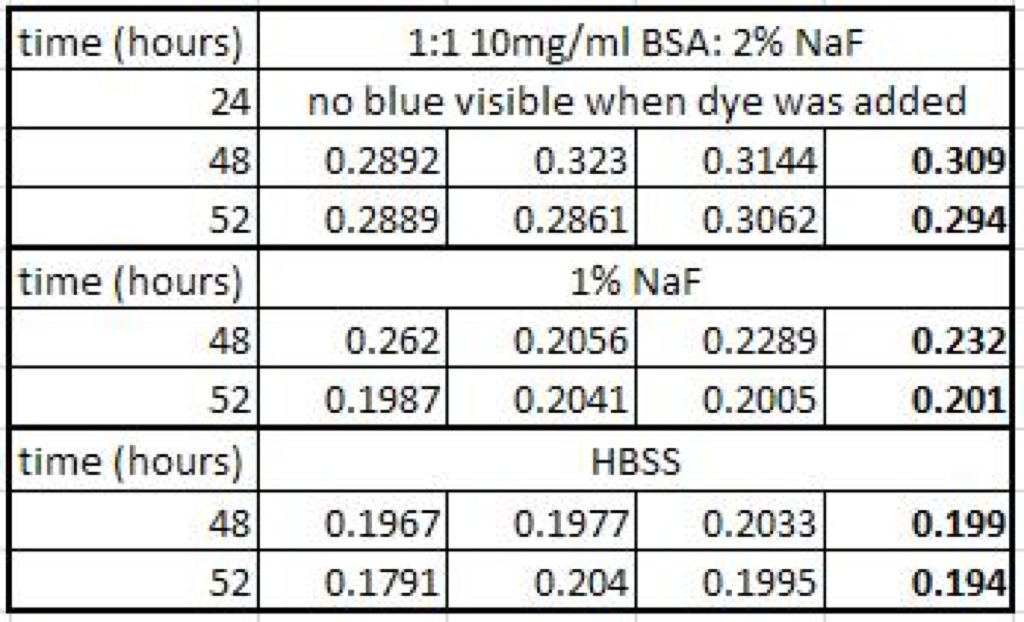
Absorbance Data:
The SepCon with BSA tested about 0.08 abs higher than the straight NaF afer 48 hours. This would be promising, except the absorbance didn’t increase at all during the next 24 hours. It is possible that some of the BSA splashed out of the SepCon after I did the 24 hour test. I am going to let the experiment sit over the long weekend and see if there are any changes on Monday.
UPDATE!
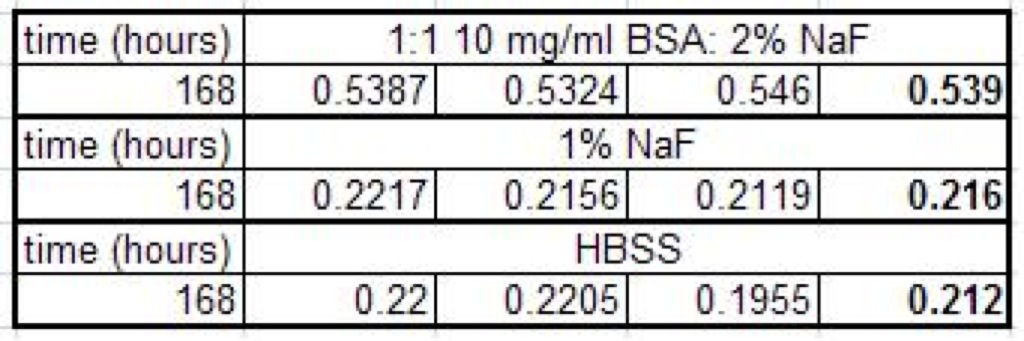
7/6/2009: Protein Assay with Fluorescine, after 7 days
When I did the Bradford assay this time, the BSA wells were decidedly blue. Quantitatively, the absorption readings are now much higher than they were last Thursday. Because of this, I would say that it is likely that the BSA is actually going through this time. It is entirely possible that I splashed more of the apical solution into the filtrate so I guess another test will be required before I can say that the BSA went through for sure. But, all in all, these are prtty promising results.
Are you performing all these tests under diffusion? Maybe you could try the pressure cell setup or in a centrifuge tube. This will speed up your tests also and may give you better results.