Side-dependent barrier function
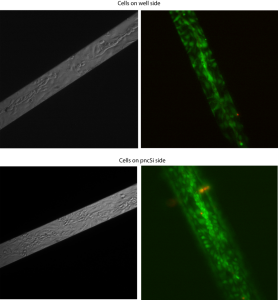
I’ve suspected that the barrier function of endothelial cells might be surface-dependent (that is, cells grown on the pnc-Si side vs. cells grown on the well side). I hypothesized that the interface between membrane and well walls would alter the continuity of the endothelial monolayer when cells are grown on the well wide. To test this, I assembled Sepcons with either the well side or the pnc-Si side facing up into the apical well of the transwell setup. I then seeded 50,000 cells/cm2 of P16 bEnd3 in the apical well, measured TEER for a week and then stained the cells with Live/Dead on the 7th day. The images are 20X and are taken through the membrane (since the cells are growing in the apical well of the setup).
Below are phase and fluorescence images of cells on 4 different samples:
There doesn’t seem to be any drastic difference in cell morphology or viability. Remember that the cells are in the apical well, so you can only see the cells on the free-standing membrane. There are tube-like structures running down the middle of the membrane in both cases. I think the bottom right image is bad-looking because of poor washing out of the dye.
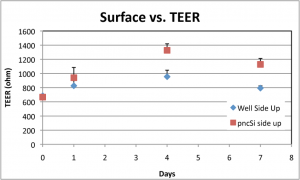
Here’s the TEER:
Each data point is from at least a triplicate measurement. TEER is much higher when the cells are grown on the pnc-Si side, as I expected. So, even though the cells grow similarly on both sides, there is a functional difference between these cell monolayers. Based on this result, I am growing the endothelial monolayer on the pnc-Si side and the glial cells on the well side since glial cells don’t form/need a continuous monolayer to function properly.
The tubes are very exciting and more evidence that the the structures over the membranes are vacuoles. Can you confirm that you did not see the tube structures (or other 3-D structures) off the membranes?
I wonder if we made membranes with widths equal to the width of the tubes if we could ‘pattern’ microvasculature on pnc-Si. If so, we might grow microvessels on pnc-Si, release them somehow, and then embed them into matrix-based tissue constructs!
We’ve got to get a handle on what about the membrane is inducing this: porosity or mechanics or something else.
Jim – I can confirm that these structures aren’t on the supported membranes – but I don’t always take images far away from the membrane. I’ve never noticed any tube-like structures off the membrane though. I’ve also never noticed them on PET membranes.
I talked to Dave and JP about making new masks to pattern cells. The resolution is pretty bad down at the single cell level, but I think slightly bigger slits (25-50um maybe) would be reasonable.
Another post dealing with porosity vs. cell morphology is coming soon.