Permeability causes vacuole formation (2)
In my last post on this subject, I showed that cutting off permeability of pnc-Si chips inhibits vacuole formation in endothelial cells. To do this, I seeded cells on the membrane side, allowed them to attach, inverted them onto a media bubble contained within a cloning ring, wet the well-side with media and then placed a coverslip over the wells. The control was to simply put a bolus of media on the well-wide. This set-up limits the volume on 1 side of the membrane to ~100nL per membrane window but the mechanics of the membranes in either set-up should be the same. Therefore, this experiment really investigates whether the difference in membrane mechanics or the difference in permeability between supported and free-standing pnc-Si drives vacuole formation. I’ve been painstakingly optimizing this protocol, and I finally got a repeat experiment in.
This sample had about 200uL media on the pnc-Si side and about 75uL of media on the well side. I used this to mimic a transwell. Vacuoles are clearly present. I only had 1 sample of this set-up and the darker areas at the edges are from photobleaching, somehow.
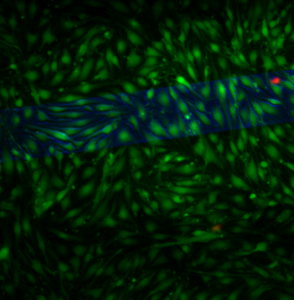
This sample was blocked with a coverslip and there are on vacuoles present over the free-standing membrane (phase channel shown in blue).
Thus, this is additional evidence that permeability and not mechanics allows vacuoles to form on free-standing pnc-Si.
Here’s another way to look at this – at 4X with normal pnc-Si transwells, you can clearly tell where the membrane is due to brighter staining and more cells.
On the sample in which the wells were blocked by a coverslip, you can’t see where the membranes are.

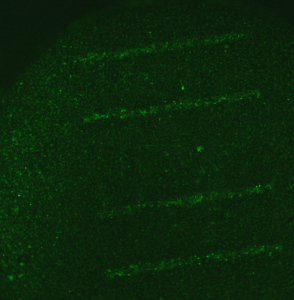

So much good stuff going on here. I think the last bit establishes that the increased fluorescence over the membranes isn’t due to lower adsorption of fluorescence over the membranes. I guess its unlikely that its due to twice the dose either since the back was blocked during staining (right). So the increased fluorescence is really due to more (and thicker) cellular material over the membranes.
Yep, you’re right. For the last (at least 6 months or so), I’ve only ever stained cells on the cell side. So, cells are never exposed to staining solution from both sides of the membrane. There are more cells and maybe more active cells.