Neurofilament H Immunofluorescence
For my BBB project, I’ve wanted to get nice images of glial cells and endothelial cells on opposite sides of pnc-Si membranes to demonstrate 1) optical transparency 2) that both cell types are in focus (and thus right on top of each other). I’ve gotten some images like that in phase contrast but I also want a 2-color fluorescent image. To do this, I need to label glial cells and endothelial cells with different antibodies that recognize cell type-specific proteins and then label those antibodies with color-conjugated secondary antibodies. For endothelial cells, I’m using VEGF and green secondary antibodies (see here). For glial cells, I’m focusing on neurofilament-H – a neural marker with red secondaries.
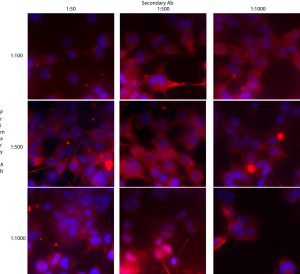
The goal of this experiment was to optimize the Ab labeling concentrations. I followed this protocol.
At 20X, all cells are brightly labeled red with blue counter-stained nuclei:
At 100X:
I tested 3 dilutions of primary and secondary antibodies. It looks like 1:100 primary and 1:500 secondary is about the best. If I find that I’m burning through this antibody too quickly, I’ll switch down to 1:500 for the primary but stay at 1:500 for the secondary.
Now I have a 2-color immunofluorescent protocol for labeling co-cultured cells. The 2-color image will be coming soon (hopefully).