If you do any separations with ultrathin nanoporous membranes please read this…
I’m not sure how many people in the current group have read all the papers that we have published, but a critical one was a JACS paper from Shigeru Amemiya where he used his Scanning ElectroChemical Microscopy (SECM) to probe the permeability of our membranes at the micron scale. In the supplement to this paper, there is an important section on wetting pnc-Si membranes. Here is the excerpt:
“Membrane Pretreatment: A membrane was treated for 1 min using a UV ozone cleaner (UVTC- 110, BioForce Nanoscience, Ames, IA), and rinsed with isopropyl alcohol for 3 minutes. The membrane that was thoroughly wet with isopropanol was placed in the SECM cell filled with an electrolyte solution containing redox molecules (Scheme S1). The wet membrane surface was protected from air, which readily blocks nanopores. For instance, the tip current in a line scan above a membrane dried after the cleaning processes was very low and non-uniform (black line in Figure S2a – 2nd figure at the bottom of post), indicating that the membrane surface was covered with air bubbles to hinder Ru(NH3)6 3+ transport. A larger, nonuniform response was also observed with a membrane (red line in Figure S2a and an image in Figure S2b), which was not adequately wet with isopropanol when being immersed in the electrolyte solution. All data except those in Figure S2 were obtained with membranes that gave a large, uniform current response to Ru(NH3)6 3+ in a line scan as shown in Figure 1b – immediately below. When needed, an electrolyte solution was exchanged with another electrolyte solution by repeatedly removing the original solution and adding the new solution so that the membrane surface was not exposed to air.” Feel free to read the whole paper, as it’s probably on this server somewhere, otherwise just search for Amemiya and McGrath and it will pop up.
Here is how a permeability scan should look (b) and some basics on the technique:
Here is what happens when the membranes are not carefully wetted, in Shigeru’s experiments – they had to do an ozone clean immediately prior to wetting, followed by a soak in IPA for 3 minutes, followed by careful displacement of the IPA with buffer:
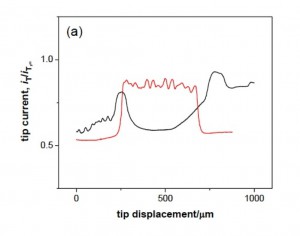
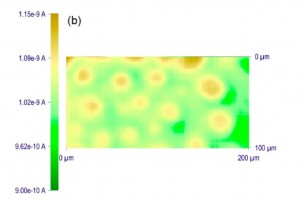
The figure to the left above shows what happens when you just wet a dry membrane with buffer (black line). It’s hard to interpret the magnitudes here, but the effective porosity is very very low. The image on the right is a 2D scan showing of inhomogeneous the permeability of a poorly wetted membrane can be. Interestingly, in diffusion our ability to stir is so limited that even a very low porosity membrane with blocked pores may still show reasonable transport. However, convective flow like in a sepcon or a pressure cell will show very low flow if the pores not efficiently wetted. I think we have a few surface treatments that have been effective in the past, but I’m not sure if anyone still uses them.
Please keep these graphs in mind when you run your experiments. Also, realize that there are NO proteins in any of these solutions. If a protein laden solution were ever to come into contact with a dry membrane (this is nearly criminal), the membrane surface will behave like fly paper and become covered with all kinds of denatured proteins, in a highly variable way. Just remember when you are working with an experimental nanomaterial, you cannot take anything for granted….
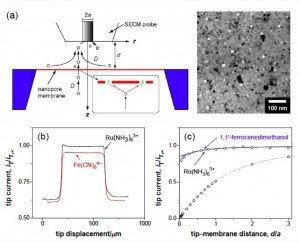
The full paper that Chris refers to is found here:
https://trace-bmps.org/groups/journalclub/wiki/a6a4c/NRG.html