Reduced diffusion distance increases rate of solute transfer through membranes
See my previous post for background information.
It has been established that diffusion of small particles (diameter < roughly 1/3 pore diameter) across ultrathin membranes is mediated almost entirely by the ability of the particle to discover a pore, rather than by its ability to diffuse through a pore once one has been found. In turn, the ability of a solute to discover a pore is intuitively mediated by the likelihood that it will be close to the membrane at any given time, which is increased when the average distance between a solute and the membrane is reduced. Thus, reduction in gasket height in diffusion experiments is expected to increase the speed with which the system approaches equilibrium, and this has already been demonstrated to be true in my previous post.
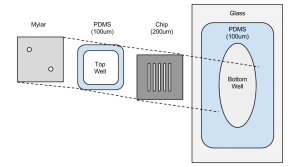
Previously, however, reduction in gasket height below 300um was shown to be only marginally effective in increasing the sieving coefficient, not living up to theoretical expectations. As explained in an edit made to my previous post, this problem arose due to the surface tension of the water in the top chamber (in that experiment, the retentate) causing the solution to prefer a droplet conformation over conforming to the boundaries of the gasket, as occurred in previous experiments with larger gaskets. In order to fix this, Jim suggested a lid be added to the system to hold the solution down in the well, and constraining it to 100um in height.
This design is identical to the previously employed design except for the mylar lid placed on top of the top gasket. Two holes are cut out of it to allow simultaneous flow of solution in and air out, or vice-a-versa. The chip is oriented with its membrane surface facing down into the bottom well.
In addition to this modification, an attempt was made to investigate the effect of diffusion direction on diffusion time. The five-slot SEPCON chip used here has a height (and thus a well depth) of 200um, twice as thick as the gaskets. This has the effect of increasing the effective diffusion length from the top well to the bottom well to 300um total, while the bottom well to the top well remains at 100um. In order to test the effect of this, two covered experiments were run, one with the filtrate on top and retentate on the bottom, and the other with their roles reversed.
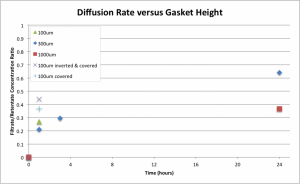
The covered and inverted (i.e., filtrate on top) system was dramatically better than the previous, uncovered 100um system. While the old 1000um system only reached a sieving coefficient of roughly 0.37 after 24 hours, the covered and inverted system beat it at 0.44 in just one hour. The covered but not inverted system had an almost identical sieving coefficient to the 1000um 24 hour case, suggesting that the inversion of the system favors solute transfer to some degree. This probably means that the slowing effect of increasing the retentate volume (and/or changing its shape, as the top and bottom wells are not of the same shape) was less significant than the slowed filtrate-to-retentate backwards diffusion rate owing to the increased diffusion distance in the filtrate. Future efforts may seek to quantify the effect of shape and filtrate-retentate diffusion distance relationships.
As it stands, this is an excellent step forward. Rapid diffusion could dramatically improve the efficacy of filtration-based assays, especially those involving small volumes and/or concentrations.