The Sieving Coefficient for Our Membranes is Time-Dependent (modeling post)
We’ve known for some time that the sieving coefficient of our membranes changes over time. The reason for this is that at the start of the separation the concentration just above the membrane is the same as the bulk concentration, but as more and more fluid begins to pass through the membrane we get concentration polarization, and the membrane concentration increases. This means that there’s a greater concentration jump across the membrane, which means that both diffusion and convection “carry” more gold per unit of water they pass. In the extreme case, if we allowed any separation to continue for an infinitely long time, the concentration at the membrane would increase unboundedly (which is physically unrealistic – sterics will keep the concentration at the membrane from growing once the spheres are packed), and as long as some gold could get through the membrane the sieving coefficient would approach 1. For reasons of simplicity, we’ve been neglecting this time evolution, but after wrestling with the data from my last post we’ve decided to tackle it.
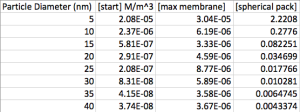
Below is the problem in one graph. We’re plotting sieving coefficients for various sizes of gold verses the amount of volume passed. The y-intercept of these lines represents the “well-stirred” case, meaning that there is no concentration polarization at all ([@membrane] = [bulk]). As more volume is passed, there is a buildup ([@membrane] > [bulk]) and a shifting of sieving coefficients.
I’ve also plotted the membrane concentration over time:
And here is a table showing that the final concentrations at the membrane are well below (5+ orders less than) the concentration of a spherical pack.
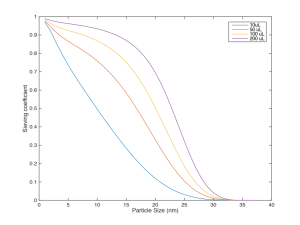
Here are some sieving curves at different volumes passed:
Jim and I have been discussing the relationship of membrane concentration to sieving coefficient for some time, and he’s asked me to explain the apparent contradiction present in the following three statements:
- Better resolution separations mean that molecules smaller than the cutoff pass more efficiently
- Better passage of small molecules should lower the concentration at the membrane
- Higher concentration behind the membrane leads to better resolutions.
I think the critical insight to answer this is to think of two different fluxes. There’s the flux into the region right above the membrane where particles are building up, and there’s the flux through the membrane. As the concentration behind the membrane grows, the flux through the membrane grows as well. But the flux through the membrane will always be less than the flux into the region right above the membrane. A concrete example:
There is a 10% chance that a gold nanoparticle 20 nm in diameter will make it through a NPN pore 37 nm in diameter.
If you take a perfectly mixed solution (so there is no concentration polarization) of this 20 nm gold and filter it with an NPN membrane with 37 nm pores, most of the gold will get rejected, and the rejected gold gets mixed back into the solution immediately. At each “step” of the separation, 90% of the gold that was in the water that passed through the membrane over the course of the step is rejected and returned to the bulk solution. Because the concentration at the membrane surface is unchanged and is the same as the bulk concentration, the sieving coefficient of each step of the separation will be 0.1, and therefore the sieving coefficient of the entire separation is 0.1, meaning 20 nm gold is above the size cutoff of the membrane.
If you turn off the hypothetical perfect mixer and do the same separation, most of the gold is still getting rejected, but now the rejected gold builds up behind the membrane. The very first “step” of the separation will have a sieving coefficient of 0.1, since the solution is well stirred at the start of the separation. But the second “step” has a higher concentration at the membrane, and that sieving coefficient of 0.1 is applied to the higher concentration.
Let’s give that some more hard numbers. We’ve fixed the pressure so that 1 uL of fluid passes through the membrane in 1 second. The bulk concentration of solution is 100 particles per 1 uL of fluid. In the first second of the separation, 1 uL of fluid passes through the membrane, and it carries 10 particles. In the well stirred case, for the 2nd second of the separation, 1 uL of fluid passes through the membrane and it carries another 10 particles, and so on for the duration of the separation.
In the dead-end/unstirred case, the 2nd second of the separation sees a higher concentration at the membrane. The 90 particles that were rejected in the first second are now just above the membrane, and though a few of them have diffused far enough away from the membrane that they won’t interrogate the membrane during the 2nd second, many of them will. The 1 uL of solution that passes through the membrane carries its original concentration of 100 part/uL, but now also has to carry some of the previous rejected gold, giving it an actual concentration of say 160 particles per uL. Since the probability of entry is still 10%, that means that 16 particles pass through the membrane.
The sieving coefficient of the two separations, after only two seconds, are given by:
S(mixed) = [filtrate]/[starting solution] = (20 particles/2 uL)/(100 particles/uL) = .1
S(unmixed) = [filtrate]/[starting solution] = (26 particles/2 uL)/(100 particles/uL) = .13