Updates on Dual-Scale Membranes (µNPN): Collagen Gel Backing and Shear Validation
Introduction
In response to recent requests by our reviewers, I am looking to address two main concerns: 1) How does a collagen gel backing affect leukocyte transmigration, and 2) How does shear influence leukocyte transmigration. While we believe these areas are rich in information and need a significant amount of focus to tackle, we do believe these questions, in a more broad sense, are key in the advancement of our dual-scale membranes. This post will address our response experiments and further expand on their potential implications going forward.
Methods
Dual-scale membranes were fabricated with micropore spacings equal to 24 um. This is the first use of such membranes; previously all membranes were fabricated in the ‘gradient’ and ‘side-by-side’ formats. For gel-backing experiments, 2 mg/ml rat tail collagen I gels were cross-linked directly in the 300 um ‘trench’. 1/6th mg/ml fibronectin was used to coating the top surface of the membrane. HUVECs were perfused into the top channel at 40,000 cells/cm2 and left to adhere and proliferate over 24 h. Freshly isolated neutrophils were added to the top channel at 4.5 million/ml and left to settle for 5 mins. 10nM fMLP in MCDB-131 (HUVEC media) was added to the bottom channel and time-lapse imaging was initiated. For shear experiments, gel-free dual-scale membranes were coated with fibronectin and seeded with HUVECs. After 24 h of static incubation, microfluidic devices were incorporated into our flow setup and flow was initiated to achieve a cell shear stress level of 4.5 dyn/cm2. After 24 h of shear priming, devices were disconnected from the flow circuit and processed for neutrophil migration experiments as described above.
Results and Discussion
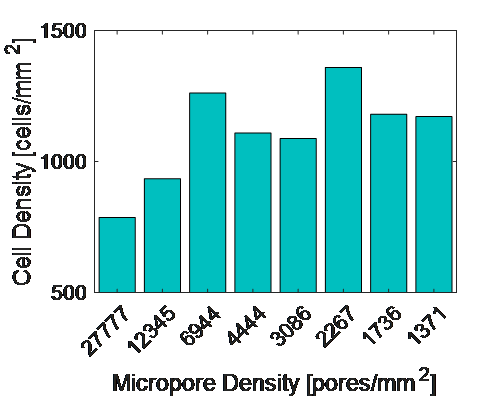
HUVEC spread area is reported to be roughly 2000 um2 (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4749947/). The following graph gives us a metric for micropores/cell on our dual-scale membranes.
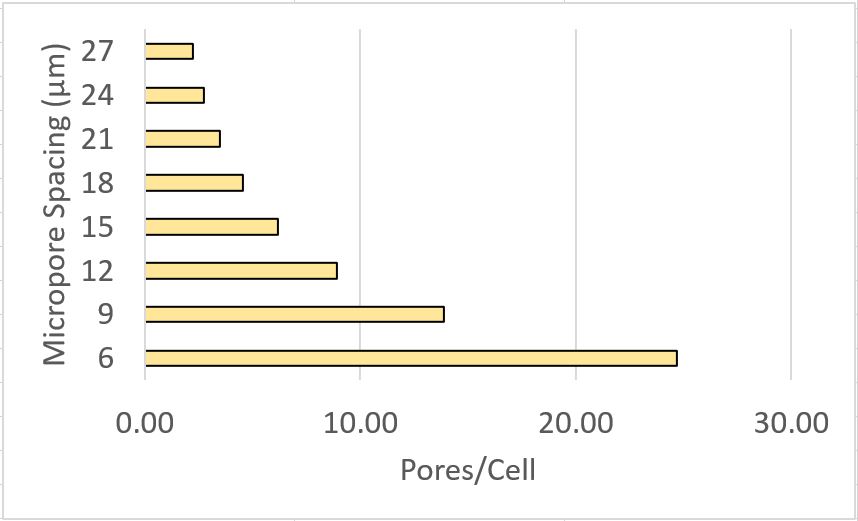
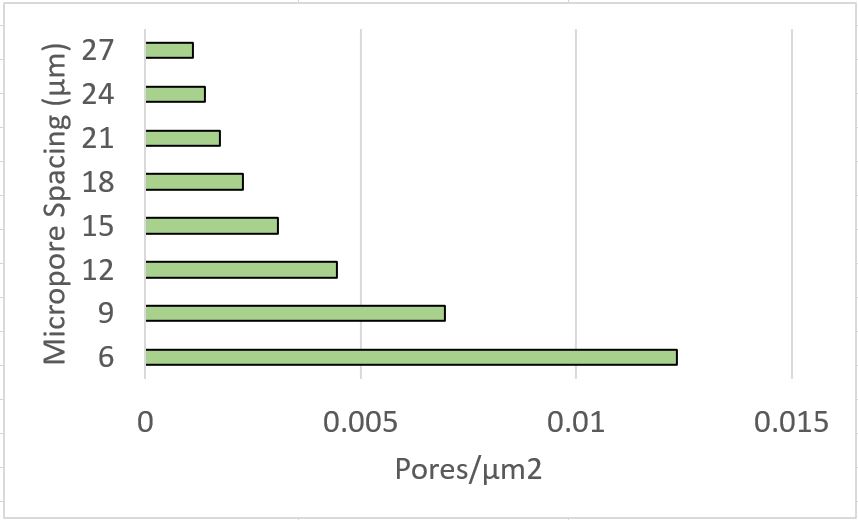
This data is interesting as it may be useful in selecting microporous membranes for shear experiments.
JK previously made me roughly 6 dual-scale membranes with pore spacings (center to center) equal to 24 um. Since this pore density showed good cell adherence and imaging in ‘gradient’ membrane experiments, these were selected for gel and shear priming experiments with leukocyte migration. As seen in Figure 1, gel backing did not limit neutrophil transendothelial migration, but did add a step of sub-membrane, extracellular matrix crawling.
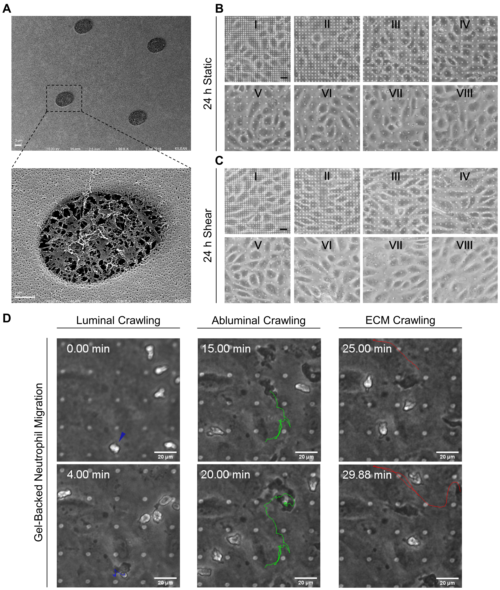
When endothelial cells were shear primed for 24 h, we observed a reduction in neutrophil transmigration, with prominent instances of reverse transmigration and probing (Figure 2).


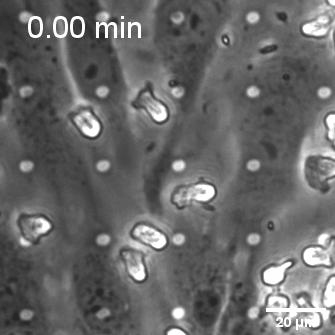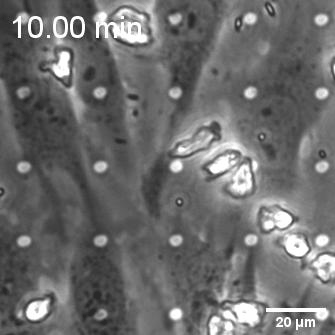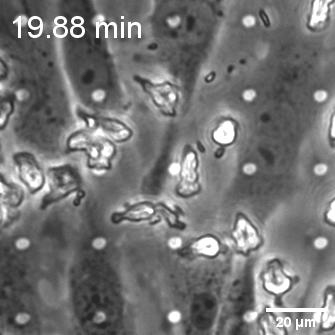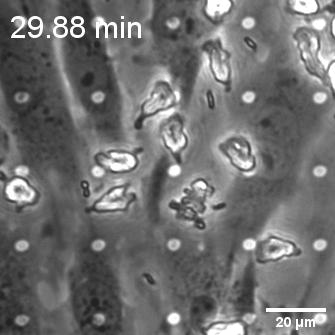
This result is not unexpected, and in a way validates our claim that shear stress enhances endothelial barrier properties. Previously Nash’s group demonstrated the net decrease in neutrophil transmigration in response to increased duration of shear stress (Figure 3; Sheikh et al., Blood 2003).

Conclusion
With these data, we will submit our revisions. Importantly for our group, I believe we can now have confidence in the use of dual-scale membranes in a variety of applications. With help from SiMPore, we hope to expand production of these membranes to a wafer scale.