Discontinuous surfaces reduce nuclear YAP localization through diminished cell spreading in ADSCs
We previously explored cellular behavior on porous substrates and discontinuous substrates. We have seen differences in cell migration and cell-substrate interactions in the form of altered f-actin polymerization, focal adhesion formation, and fibronectin fibrillogenesis on these discontinuous substrates. We were curious about mechano-sensing on these substrates. To investigate mechano-sensing on disrupted surface, we developed a non-fouling micro-patterned substrate using PLL-g-PEG similar to a previous study. Figure 1.A shows the environmental AFM of hydrated PLL-g-PEG brushes on the SiO2 substrate.

Fig.1. A. Atomic force microscopy of PEG islands on the SiO2 substrate, B. Scanning electron micrographs of SiO2 membrane with 3.0 µm diameter pores.
We first investigated the f-actin polymerization and focal adhesion formation of Adipose-derived stem cells (ADSCs) on the PEG islands. We found similar weakened cell-substrate interaction as our previous studies on this non-fouling patterned substrates. We have seen a significant reduction in F-actin polymerization and a slight but not significant decrease in focal adhesion formation.
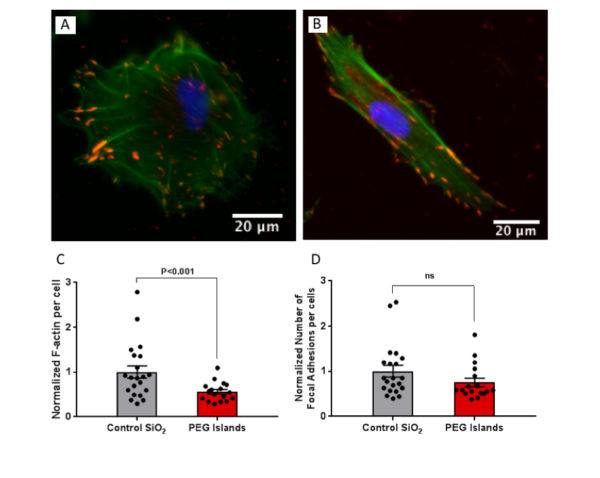
Fig.2. Fluorescence images of ADSCs stained for F-actin (phalloidin, green), focal adhesions (anti-vinculin, red) and nuclei (DAPI, blue) on (A) Control SiO2 and (B) PEG islands. (C) F-actin intensity per cell normalized to control mean value. (D) Distinct focal adhesions per cell normalized to control mean value.
Prior studies suggested that F-actin and focal adhesion do not drive changes in YAP localization and they are not strong indicators of YAP localization, but YAP localization can affect F-actin and focal adhesion formation. However, it was previously shown that by controlling cell spreading, YAP localization can be modulated even without significant decrease in focal adhesion. Overall, the dominant player in mechano-sensing was introduced to be spreading area. Therefore, we investigated cell spreading and YAP localization in the next step. We observed a significant reduction in spreading area of mesenchymal cells on these micro-patterned substrates. We looked at YAP localization after 6 h of cell seeding and found no difference between the control and PEG islands. However, after 24h cells responded to the substrate and we observed a statistically significant decrease in nuclear YAP localization on the discontinuous substrates, and it was consistent with the previous studies. In fact, we patterned micron scale non-fouling islands on a stiff substrate and we were able to reduce nuclear yap localization by reducing cell spreading.
These results show two main points. First, the surface disruption and weakened cell-substrate attachment can affect cell spreading and mechano-sensing despite the substrate being extremely stiff such as SiO2 on a silicon wafer. Second, it confirms that cell spreading plays the main role in mechano-sensing and YAP localization, and even without a significant decrease in focal adhesion, we observed a significant decrease in nuclear YAP localization.
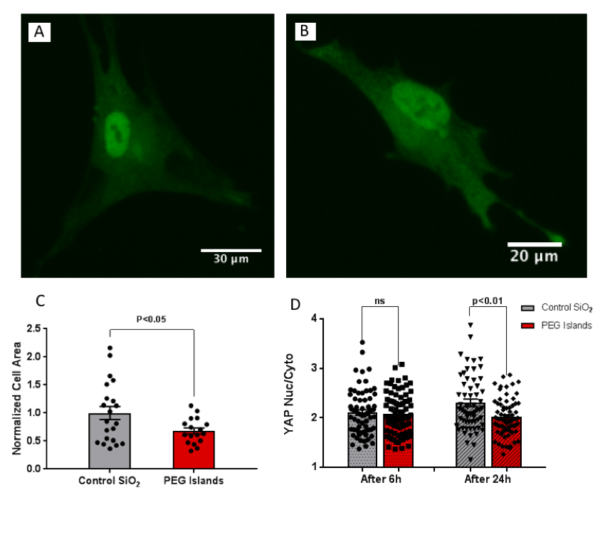
Fig.3. Fluorescence images anti-YAP on (A) Control SiO2 and (B) PEG islands. (C) Cell spread area was quantified from F-actin images. (D) YAP nuclear-cytoplasmic ratio is
quantified at 6h and 24h.
Since porous membranes are probably the most broadly applicable disrupted surfaces in cellular studies, we finished our study by evaluating cell spreading and YAP localization on porous membranes with the identical pattern. We found the same trend of reduction in cell spreading and nuclear YAP localization on the porous membrane. It suggests that mesenchymal cells respond to micro-porous membranes in a way that can impact cell signaling pathway and mechano-sensing without changing substrate stiffness, and it is experimentally significant because many people using microporous membranes in a variety of studies including co-culture systems and barrier models without considering these effects. We further repeated our experiments on sub-micron pores and obtained the same results which confirms our conclusion about the role of surface disruption in cell spreading and YAP localization.

Fig.4. Fluorescence images anti-YAP on (A) non-porous, (B) 3.0 µm pore diameter and (C) 0.5 µm pore diameter SiO 2 membranes. (D) Cell spread area was quantified from F-actin images. (E) YAP nuclear-cytoplasmic ratio is
quantified at 6h and 24h.