Electrospinning aligned polymeric fibers on parylene stencils: cell culture and pll-g-PEG coating update
A quick recap of the goals and initial experiments:
https://trace-bmps.org/electrospinning-aligned-polymeric-fibers-on-parylene-stensils/
After obtaining a reproducible optimized fiber deposition settings and an improved device design, we moved on to growing HUVECs on these devices as the next step. As previously stated, given that there is a parallel project with membrane masking of ECM with parylene stencils, we kept the exact same parameters for cell seeding (seeding density, passage number, length of experiment, stencil pore size and porosity, …).
The conditions used for these experiments were the following: 1) aligned fibers covered with 4 um pore sizes and 50% porosity, 2) covered with 8 um pore sizes and 50% porosity, 3) cells directly seeded on aligned fibers (positive control), 4) random fibers covered with 4 um pore sizes and 50% porosity (negative control) as well as TCPS.
These are the representative images and analyzed data from those experiments:
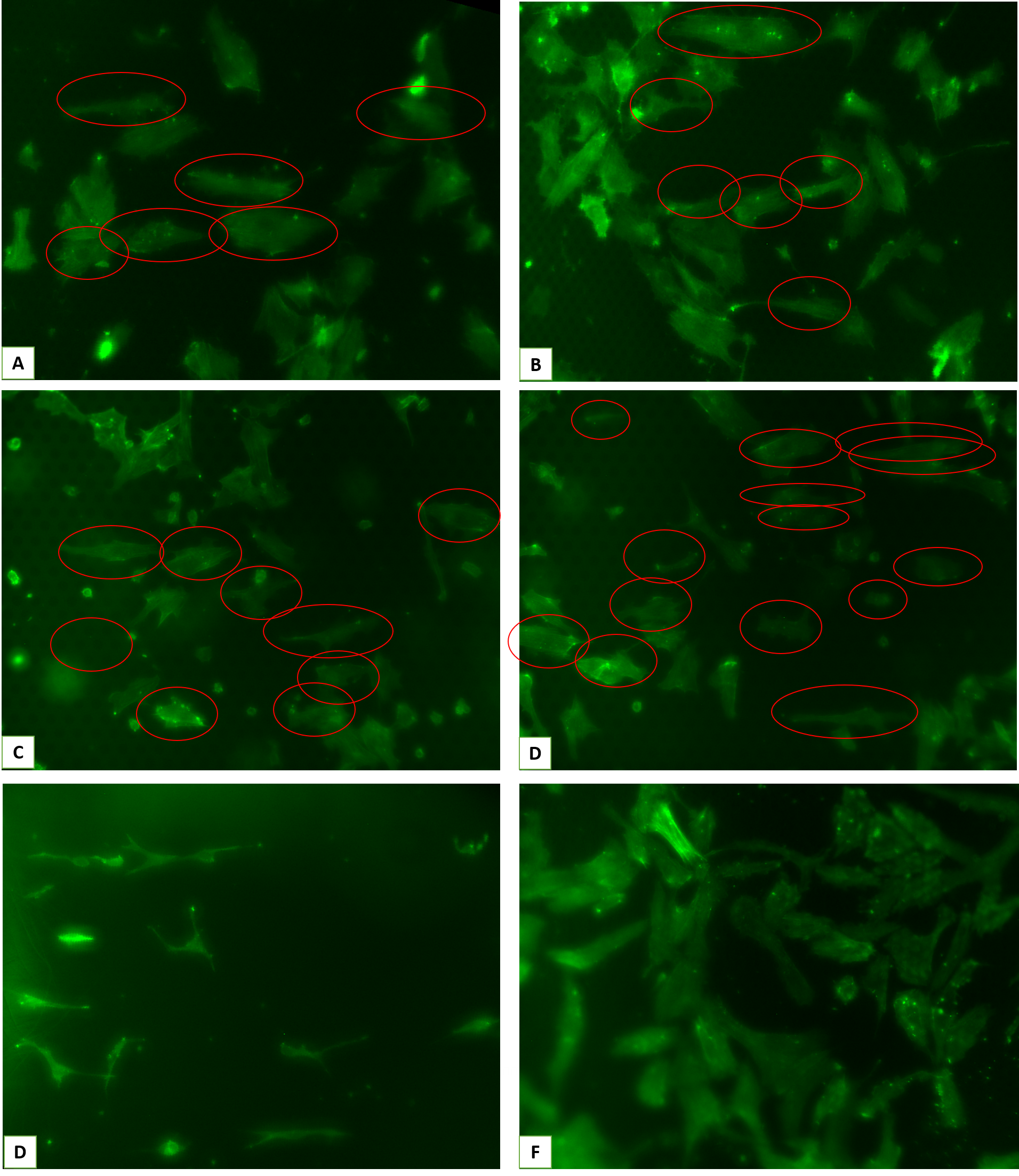
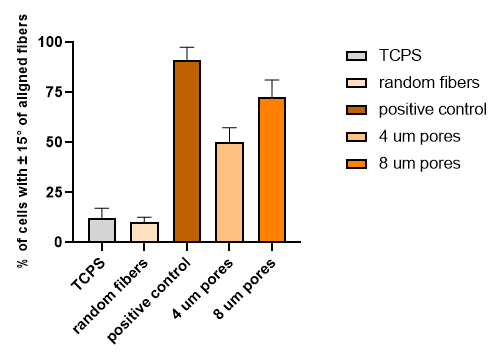
After carefully assessing the data and also considering our previous work with parylene, we expected as well as observed a disruption in cell-fiber interaction as result of HUVECs interacting with parylene even without the plasma activation, necessary for parylene C to become ideal for cell culture. In this project, we ideally don’t want any disruption like this and the whole purpose of parylene was to serve as an inert mask.
Therefore, we attempted to make the stencil non-fouling, using the protocol that Zahra developed for her ACS biomaterial science paper with Pll-g-PEG coating. Since that protocol was developed for SiO2 membranes, using recommendations and guidance from Zahra, I tested a few condition to obtain the best possible result.
Briefly, the process starts with placing pll-g-PEG in HEPES solution with a density of 0.5 mg/ml on top of the membranes for 90 minutes, followed by gentle washing. In order to visualize and quantify the effectiveness of the non-fouling coating, we coated the substrate with fluorescently labeled bovine serum albumin (f-BSA) for 60 minutes. Here are representative images from those experiments:
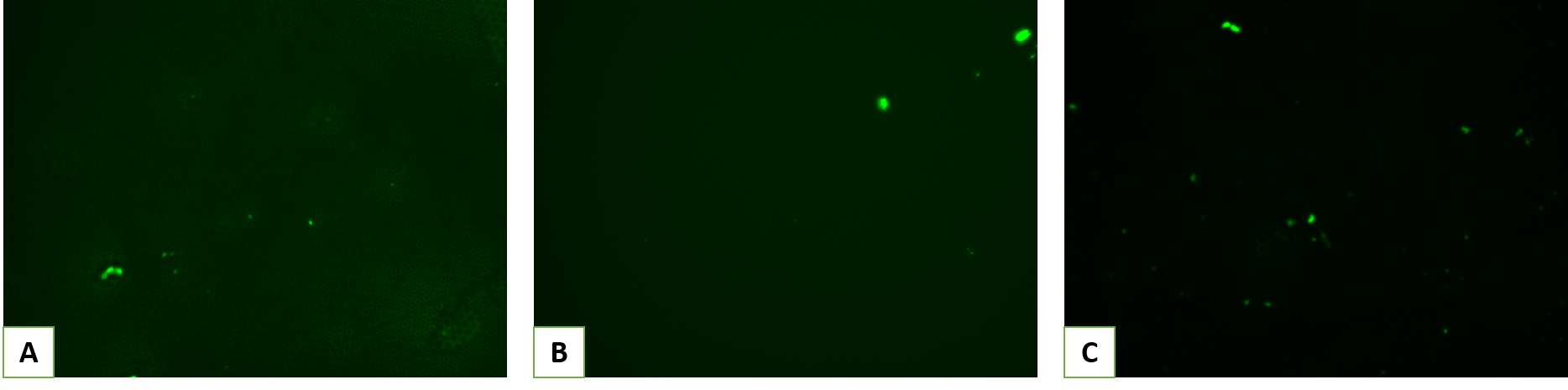
By subtracting the background noise and normalizing the intensity to non-coated samples, we obtained the following data:
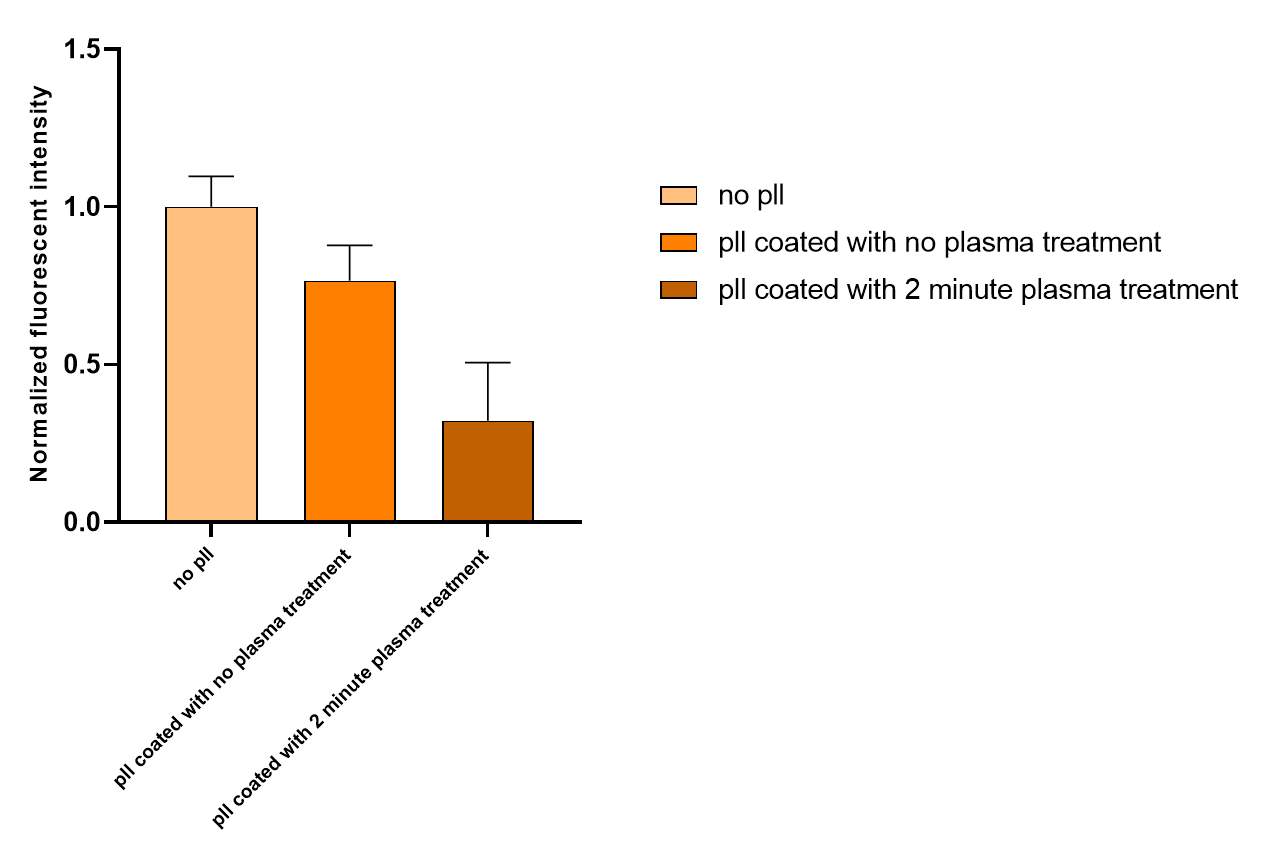
Current obstacles and future steps
Autofluorescence from fibers in GFP channel, a better design where flipping the device is more feasible, and repeating the experiments with Pll-g-PEG coated samples are among the current obstacles and future steps that we have ahead.