Effects of TNF-alpha on iPSC-EC glycocalyx (EGL)
One of the validations of the in vitro human Blood Brain Barrier system is to ensure that the endothelial cells derived from iPSC’s and cultured in our system exhibit behaviors that are consistent with endothelial cell behavior in situ. The particular aspect of BBB endothelial function addressed in this post is the expression and properties of an endothelial glycocalyx layer (EGL) and the effects of inflammatory stimulus on these layers. Literature reports have documented the existence of the EGL in multiple tissues and the elimination of the EGL as an early event after inflammatory stimulus.
In these experiments, iPSC EC’s were grown to confluence in the lower channel of an A-line device and maintained for up to 4-5 days. When shear stress was gradually (over 4-5 hours) increased to 5.6 dyn/cm2 and maintained for 44-48 hours, cells aligned in the direction of flow, and expression of both hyaluronic acid and the junctional protein ZO-1 were observed using immunofluorescence microscopy.
After two days of flow exposure, samples were tested using AFM to determine the mechanical properties and thickness of the EGL. Tipless AFM cantilevers () were fitted with a 6.0 um glass bead glued to the tip. Typically, ten repeated indentations are performed in the perinuclear region of the cell and the repeated indentations are super-imposed and averaged (to remove noise). (Cells are allowed to recover for one minute between indentations, which are done at 4.0 um/s until a force of 4.0 nN is reached.) The averaged curves are fit to a modified Hertz model of indentation of a thin elastic layer above an elastic half space. Three parameters are determined by least squares regression: The elastic modulus of the cell, the elastic modulus of the EGL, and the thickness of the EGL.
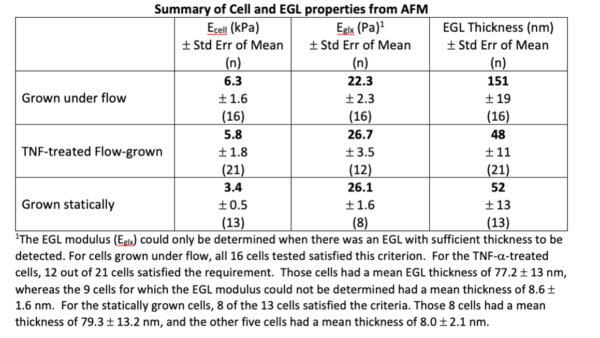
In our most recent experiments, two A-lines were seeded from a common cell culture, grown to confluence, maintained for two days after reaching confluence, then subjected to a fluid shear stress of 5.6 dyn/cm2 for 44-48 hours. One A-line was treated with TNF-a (10 ng/ml) for four hours under flow before testing and the other was tested on the same day without treatment. Results revealed little effect of TNF-a on either the cell modulus (~ 6.0 kPa) or the EGL modulus (~25 Pa), but dramatic changes in EGL thickness were observed (Figure). The mean EGL thickness was reduced from 150 nm to 50 nm, and for approximately half of the TNF-a treated cells, the thickness was less than 15 nm, so small that the EGL modulus could not be reliably determined. The results for the TNF-a treated cells closely resembled the results for cell grown statically. Out of 13 qualified measurements on cells grown statically, five showed EGL thickness less than 15 nm. The other 8 cells gave an average thickness of 79 nm, very close to the value of 77 nm for the comparable TNF-a treated cells and the overall mean thickness was ~50 nm, essentially identical to the overall average for TNF-a treated cells.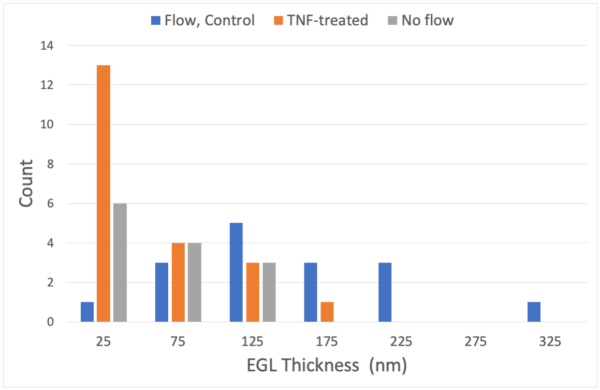
These measurements indicate that iPSC EC’s REQUIRE flow in order to develop a robust EGL, and that once this EGL is developed, the cells respond to inflammatory stimulus as expected by shedding a significant portion of the EGL.
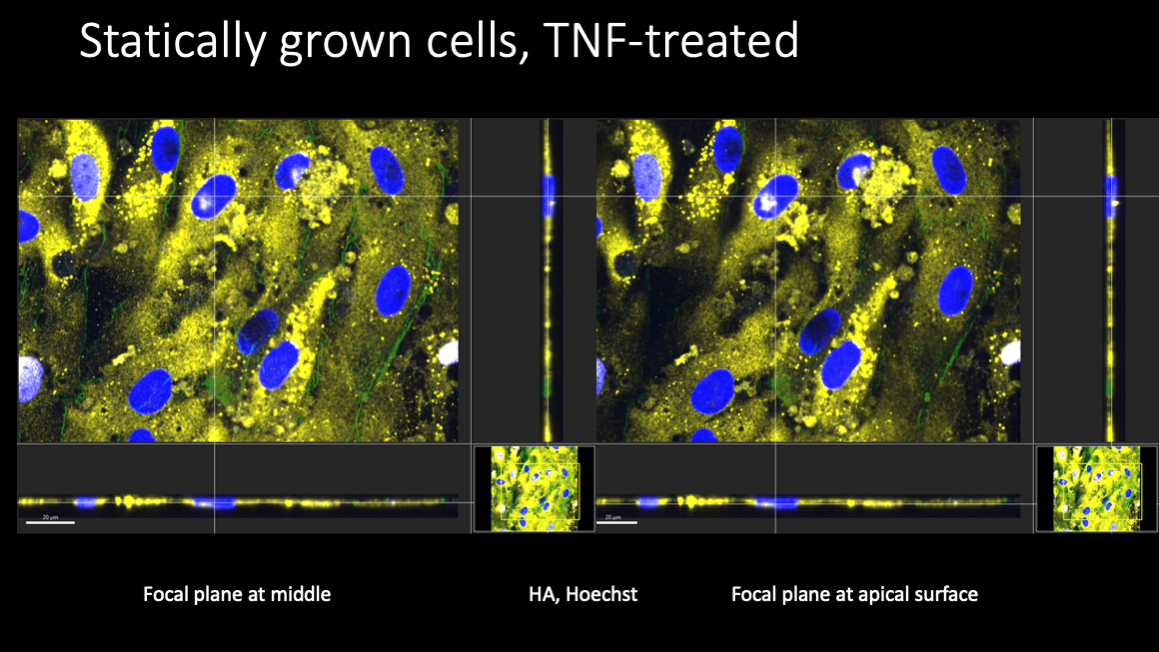
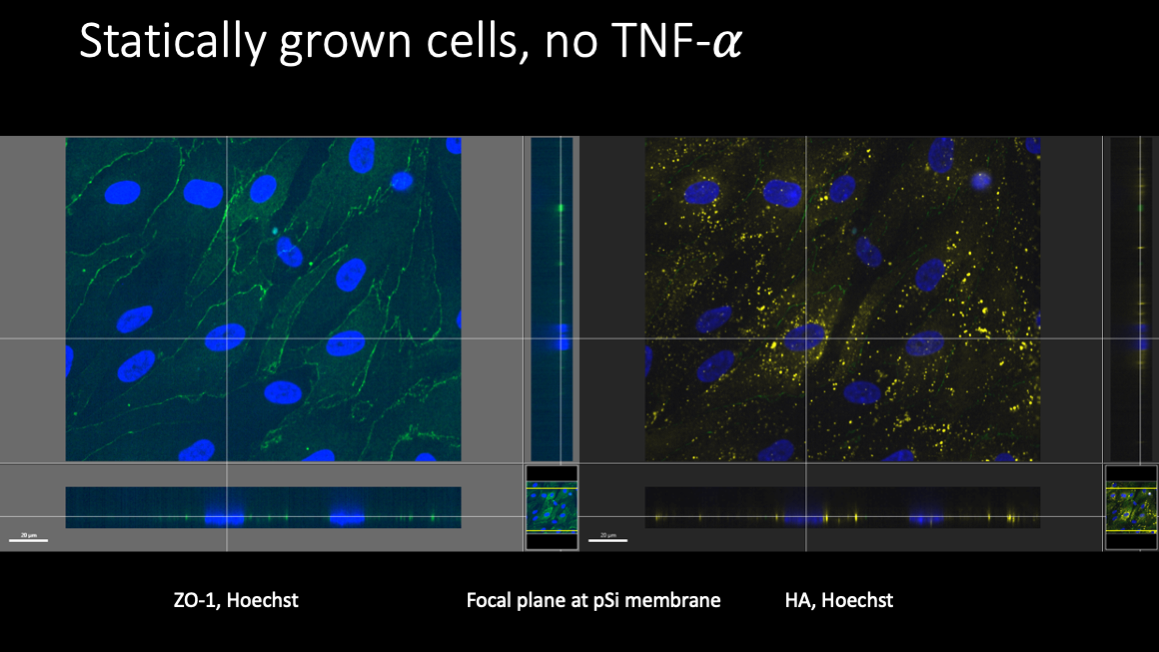
Fluorescent labeling of these cells revealed some surprises. Cells grown statically failed to produce much hyaluronic acid (HA) at all, but cell junctions were well developed.
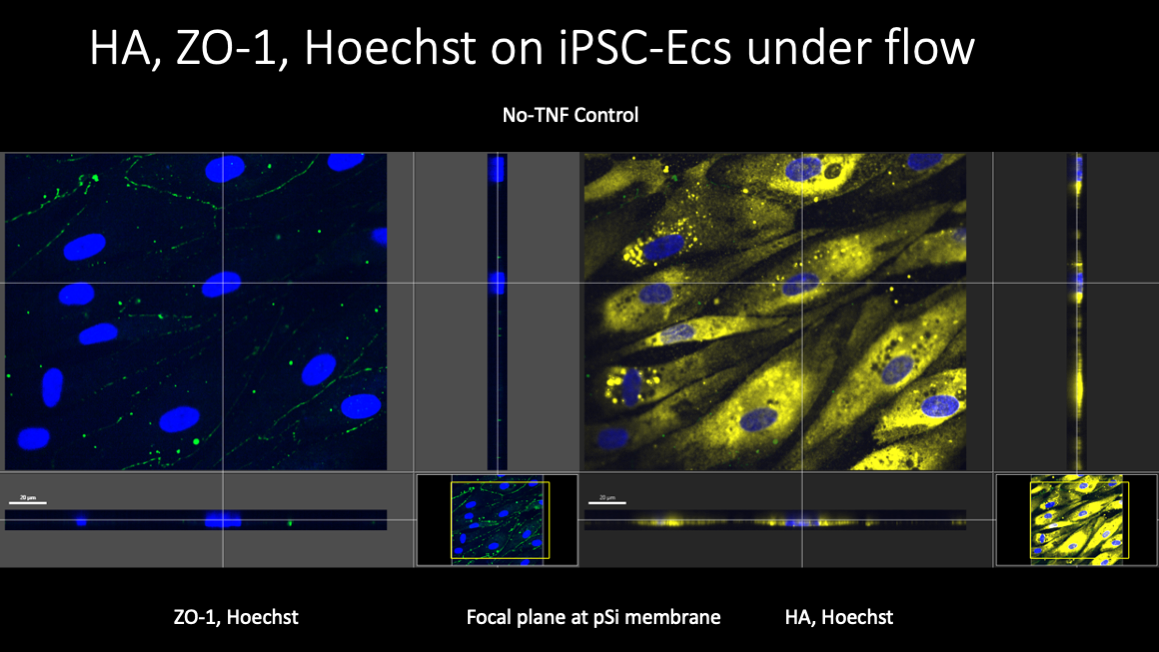
Under flow, the expression of HA increased, consistent with AFM measurements of a more robust EGL.
TNF treatment was expected to result in the elimination of the EGL, and AFM results seem to confirm this. Paradoxically, HA levels in cells grown both statically and under flow appeared to increase substantially after TNF treatment.
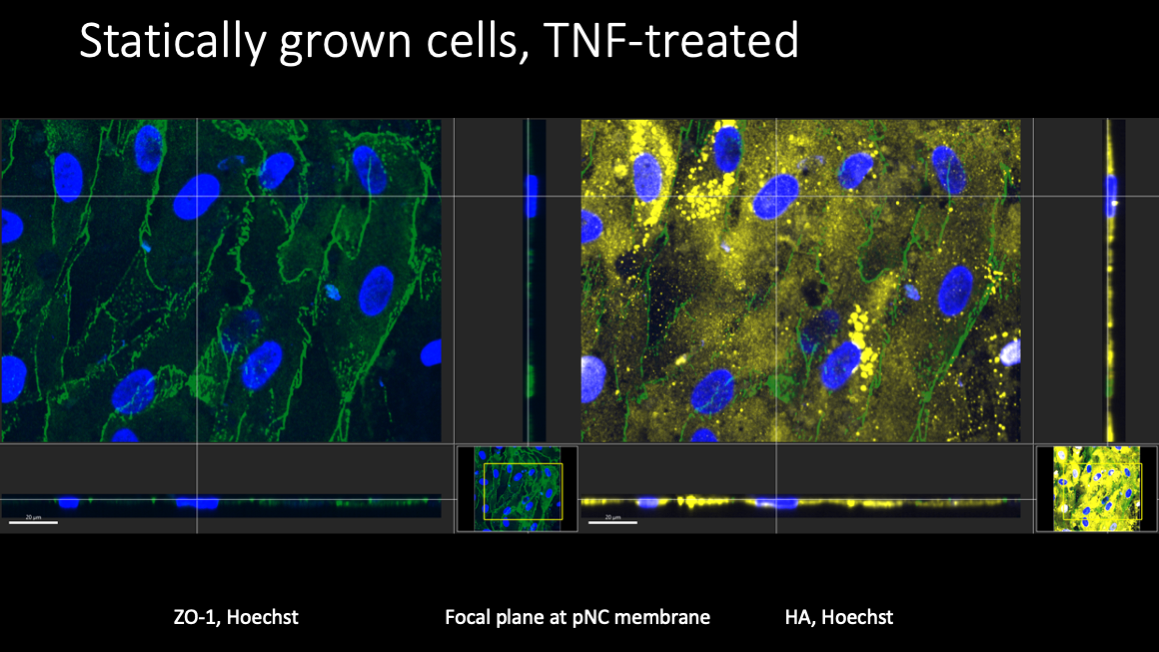
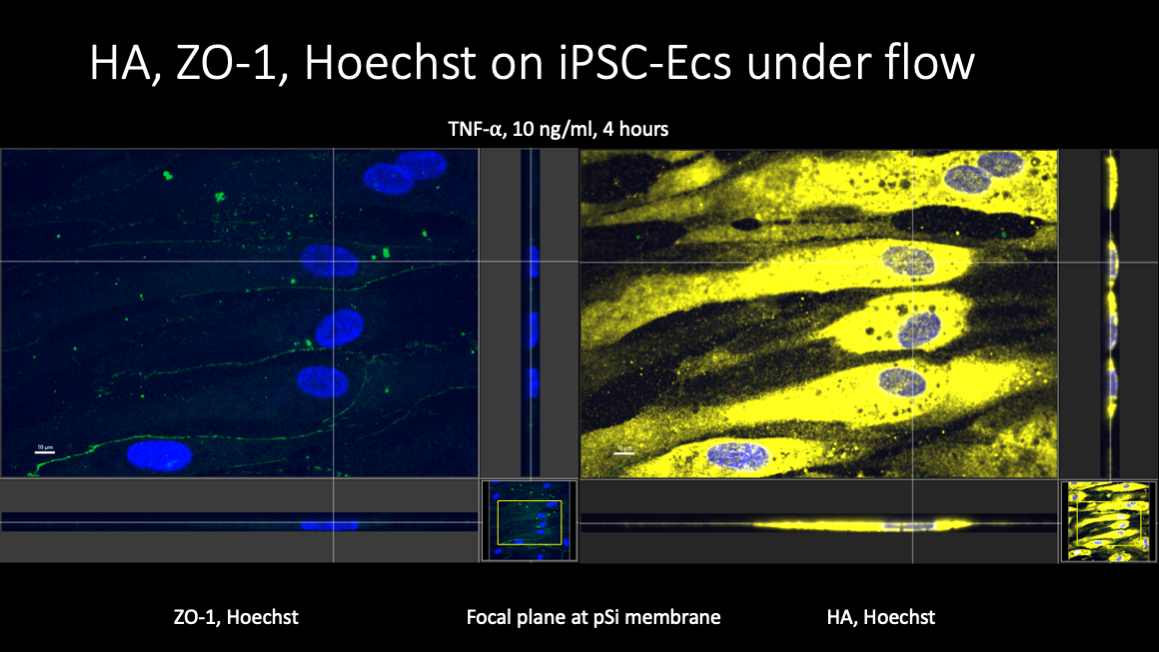
The vertical distribution of the HA was not entirely clear from the confocal images, but a substantial portion of the distribution appeared to be intracellular.