Energy needed for pore nucleation and growth
I first wrote about quantifying the pore formation process using kinetic theory last year. In this post, I will develop my thoughts a bit more.
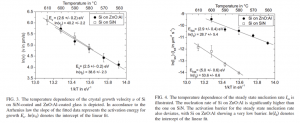
In the literature, researchers oftentimes present charts, like the two shown below, describing the Arrhenius behavior of silicon crystallization (Sontheimer APL 95 2009).
Figures 3 and 4 show the crystal growth rate (ν) and nucleation rate (Iss) as a function of temperature. Using the Arrhenius equation they extracted an activation energy for these two processes.
Using a similar method, I have plotted the pore growth rate (kp) and nucleation rate (Jp) as a function of temperature for a 15 and 30 nm pnc-Si membrane.
The activation energy for pore growth is very similar between the 15 nm and 30 nm films. This makes sense intuitively since one a pore has nucleated the growth rate should be independent of thickness since the material is the same. There is, however, a difference in the activation energy of pore nucleation. The results show that it takes 25% more energy to nucleate pores in a 30 nm film than it does a 15 nm thick film. This is reasonable given our experience with needing to anneal at higher temperatures in thicker films to “open” pores.

I’m not sure how much we can conclude about the dynamics of this process by looking at the final morphology, since many of these effects are happening during the temperature ramp. At least it’s significantly more complicated than the constant-temp growth rate in that reference.